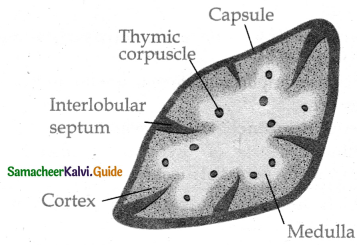
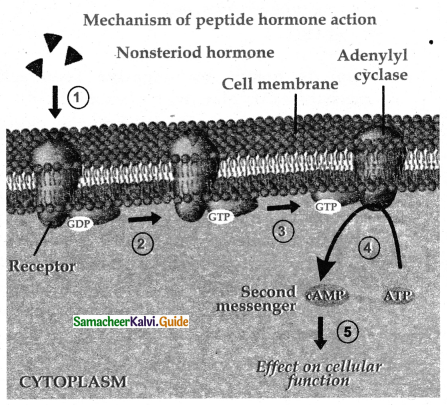
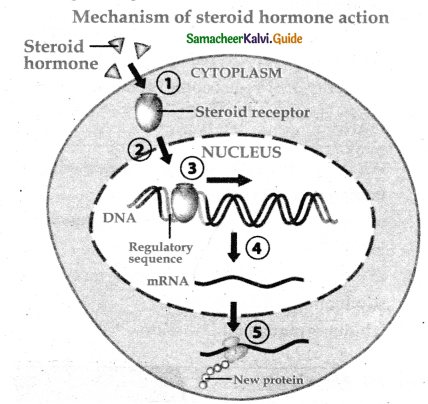
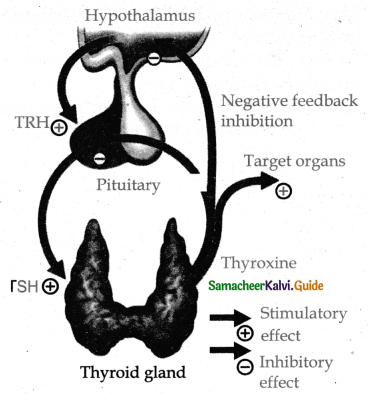
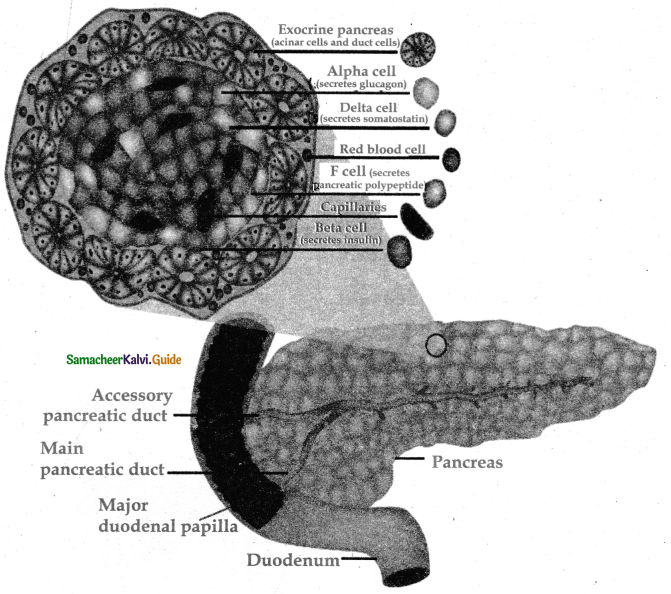
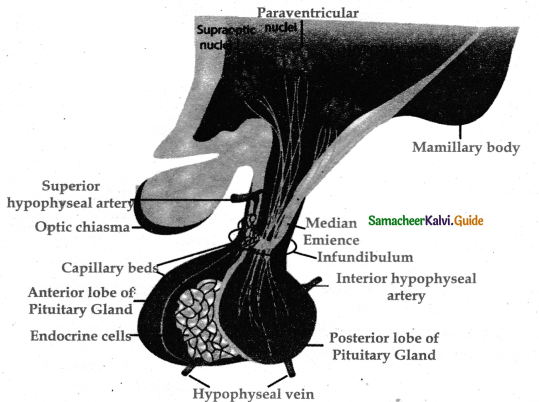
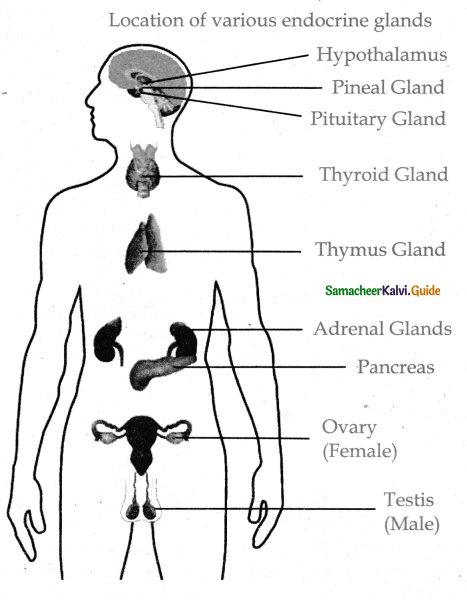
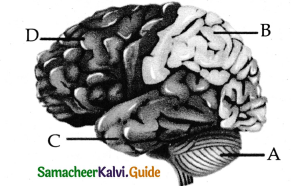
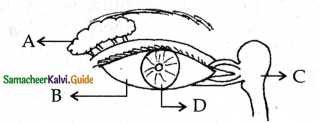
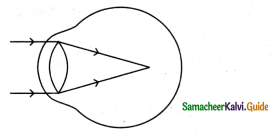
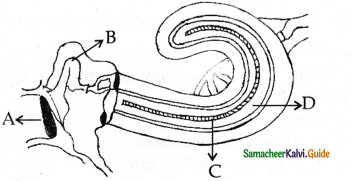
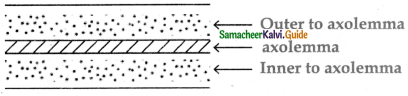
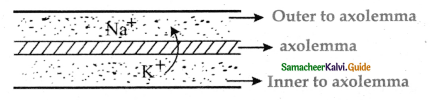
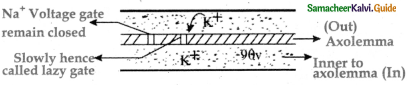
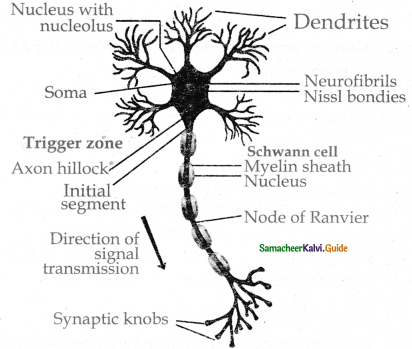
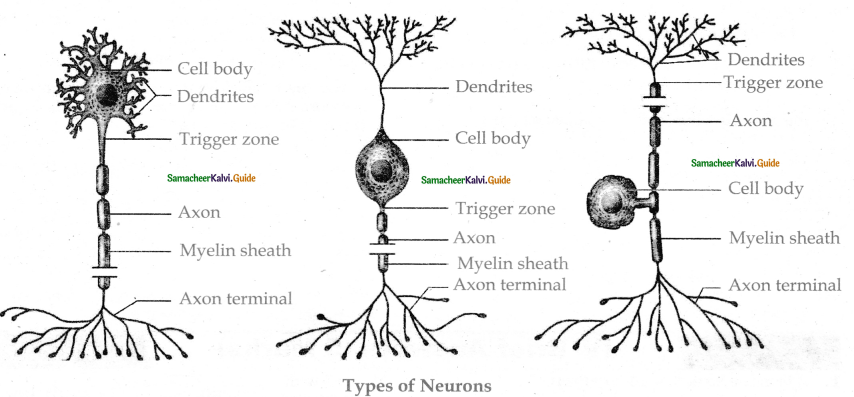
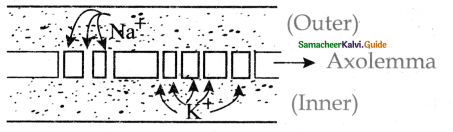
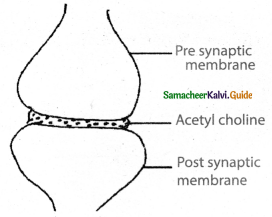
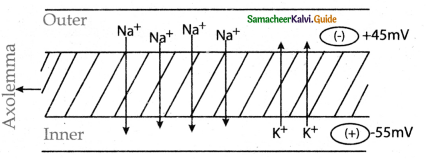
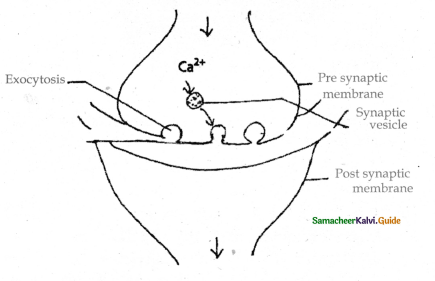
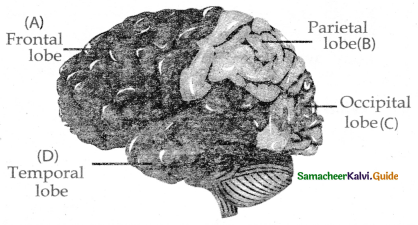
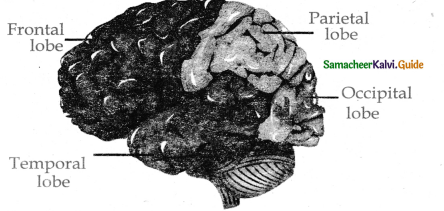
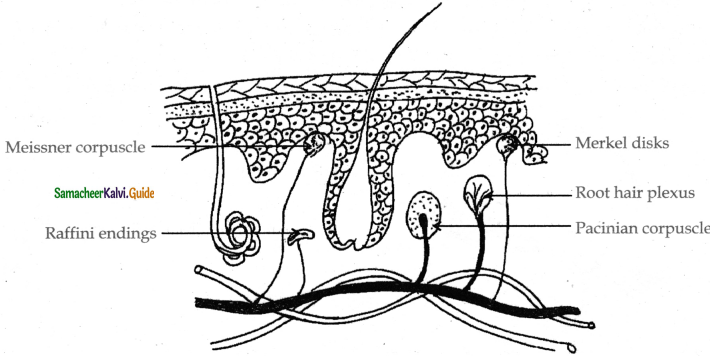
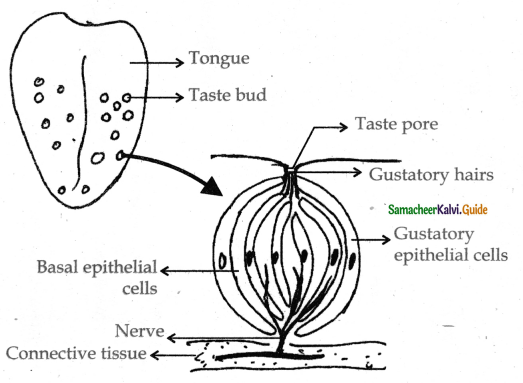
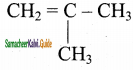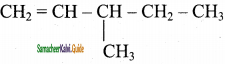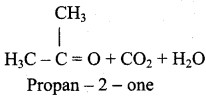Subject Matter Experts at SamacheerKalvi.Guide have created Tamilnadu State Board Samacheer Kalvi 11th Accountancy Answers Solutions Guide Pdf Free Download of Volume 1 and Volume 2 in English Medium and Tamil Medium are part of Samacheer Kalvi 11th Books Solutions.
Let us look at these TN Board Samacheer Kalvi 11th Std Accountancy Guide Pdf of Text Book Back Questions and Answers, Notes, Chapter Wise Important Questions, Model Question Papers with Answers, Study Material, Question Bank, Formulas and revise our understanding of the subject.
Students can also read Tamil Nadu 11th Accountancy Model Question Papers 2020-2021 English & Tamil Medium.
Samacheer Kalvi 11th Accountancy Book Solutions Answers Guide
Samacheer Kalvi 11th Accountancy Book Back Answers
Tamilnadu State Board Samacheer Kalvi 11th Accountancy Book Back Answers Solutions Guide.
- Chapter 1 Introduction to Accounting
- Chapter 2 Conceptual Framework of Accounting
- Chapter 3 Books of Prime Entry
- Chapter 4 Ledger
- Chapter 5 Trial Balance
- Chapter 6 Subsidiary Books – I
- Chapter 7 Subsidiary Books – II
- Chapter 8 Bank Reconciliation Statement
- Chapter 9 Rectification of Errors
- Chapter 10 Depreciation Accounting
- Chapter 11 Capital and Revenue Transactions
- Chapter 12 Final Accounts of Sole Proprietors – I
- Chapter 13 Final Accounts of Sole Proprietors – II
- Chapter 14 Computerised Accounting
We hope these Tamilnadu State Board Class 11th Accountancy Book Solutions Answers Guide Volume 1 and Volume 2 Pdf Free Download in English Medium and Tamil Medium will help you get through your subjective questions in the exam.
Let us know if you have any concerns regarding TN State Board New Syllabus Samacheer Kalvi 11th Standard Accountancy Guide Pdf Text Book Back Questions and Answers, Notes, Chapter Wise Important Questions, Model Question Papers with Answers, Study Material, Question Bank, Formulas, drop a comment below and we will get back to you as soon as possible.

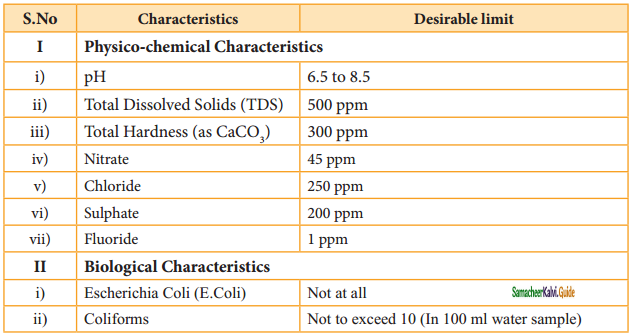
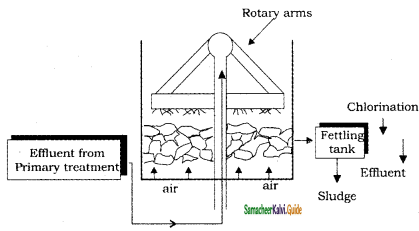
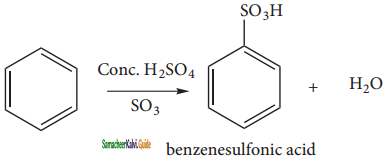
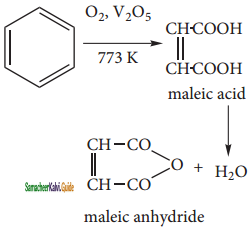
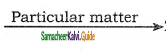 2SO3
2SO3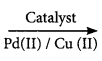 CH3CHO
CH3CHO is
is X is ______.
X is ______.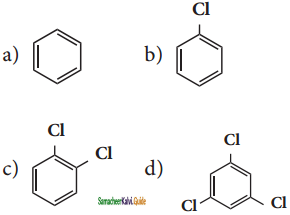
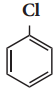




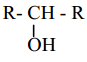


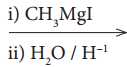 X, X is
X, X is

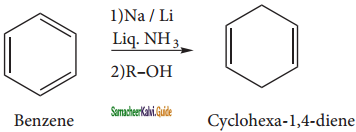
 C6H5 – C6H5 + 2NaCl
C6H5 – C6H5 + 2NaCl
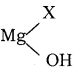


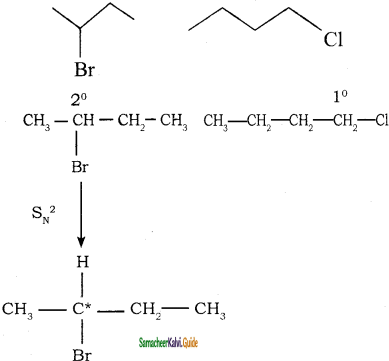
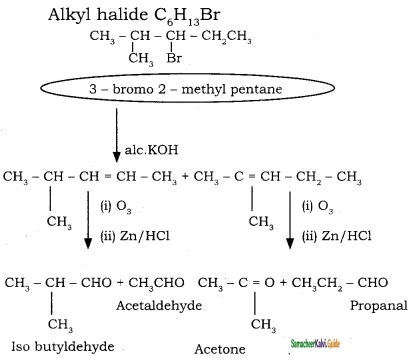
 → 2 – bromo pentane
→ 2 – bromo pentane → 3 – bromo pentane
→ 3 – bromo pentane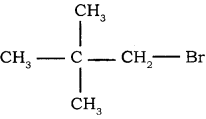 → 1 – bromo 2, 2 – dimethyl propane
→ 1 – bromo 2, 2 – dimethyl propane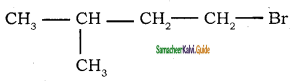 → 1 – bromo 3 – methyl butane
→ 1 – bromo 3 – methyl butane → 2 – bromo 3 – methyl butane
→ 2 – bromo 3 – methyl butane → 2 – bromo 2 – methyl butane
→ 2 – bromo 2 – methyl butane → 1 – bromo 2- methyl butane
→ 1 – bromo 2- methyl butane → (2S) – 1 – bromo 2 – methyl butane
→ (2S) – 1 – bromo 2 – methyl butane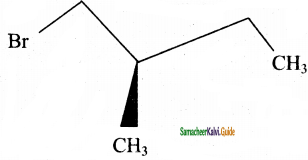 → (2R) – 1 – bromo 2 – methyl butane
→ (2R) – 1 – bromo 2 – methyl butane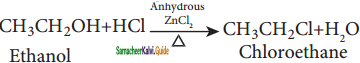



 C6H5NH2 + NH4Cl
C6H5NH2 + NH4Cl


 CCl4 + S2Cl2
CCl4 + S2Cl2







 C6H5 – C6H5 + 2NaCl
C6H5 – C6H5 + 2NaCl 2 HCl + CCl2F2
2 HCl + CCl2F2
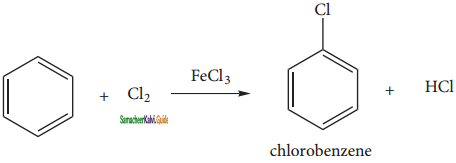
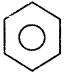 + Cl2
+ Cl2  B, B is
B, B is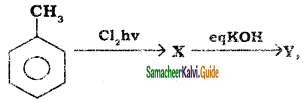 Y is
Y is

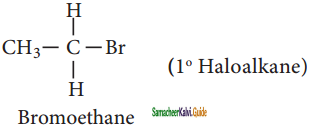
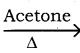 CH3CH2I + NaI
CH3CH2I + NaI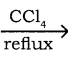 CH3CH2Br + CO2 + AgBr
CH3CH2Br + CO2 + AgBr


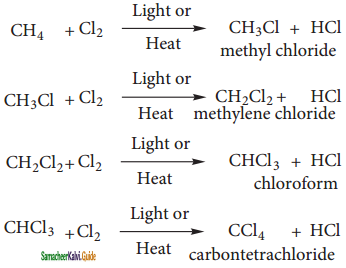
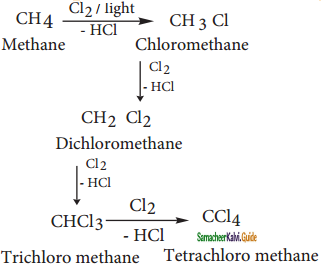
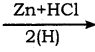 CH2Cl (Methylene chloride) + HCl
CH2Cl (Methylene chloride) + HCl CH3Cl
CH3Cl 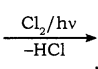 CH2Cl2
CH2Cl2 CHCl3 (chloroform) + HCl
CHCl3 (chloroform) + HCl


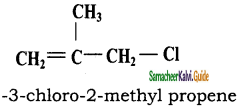
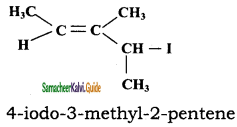




 CH3CH2SH + NaBr
CH3CH2SH + NaBr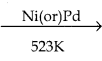 CH3 – CH3 + HBr
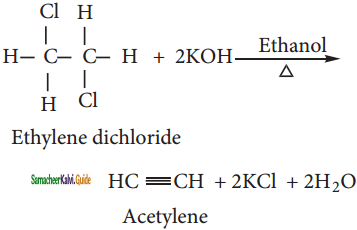
CH3 – CH3 + HBr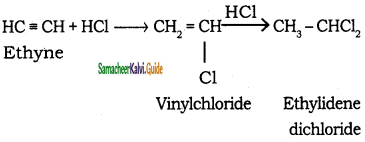



 C6H6 + HCl
C6H6 + HCl C6H5MgCl (Phenyl magnesium chloride)
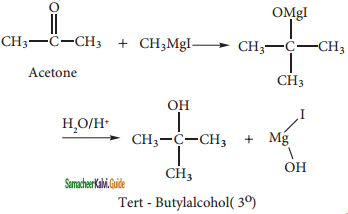
C6H5MgCl (Phenyl magnesium chloride)


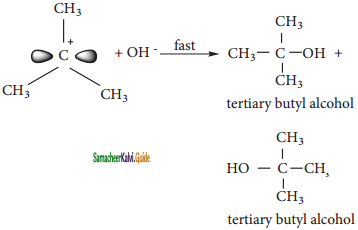



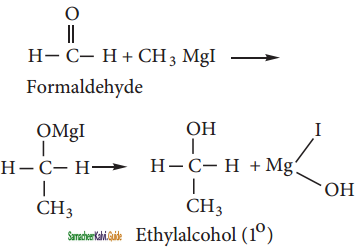





 CH2Cl2 (Methylene chloride)+ Cl2
CH2Cl2 (Methylene chloride)+ Cl2

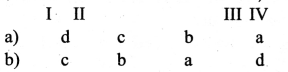






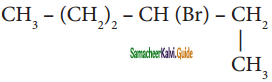
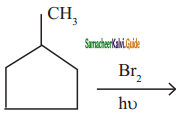
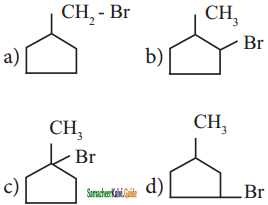
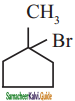
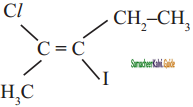
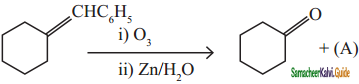
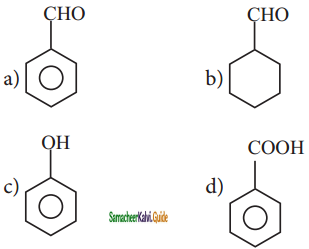
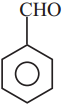
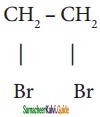
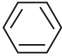
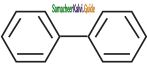
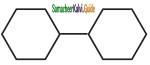
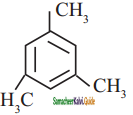
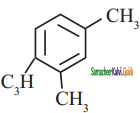
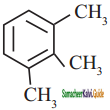
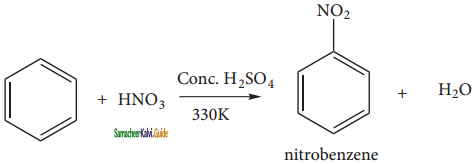
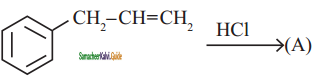 is
is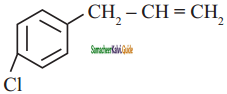
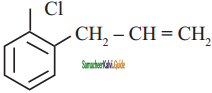
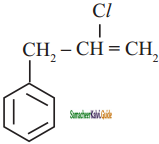
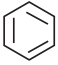
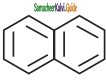
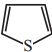
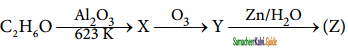
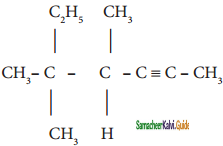
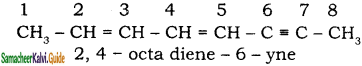
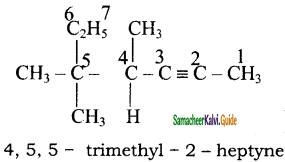
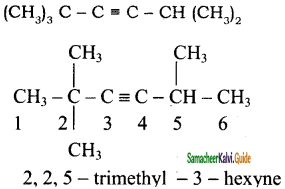
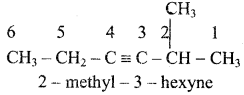
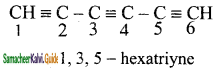
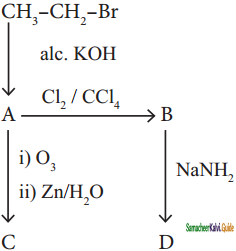
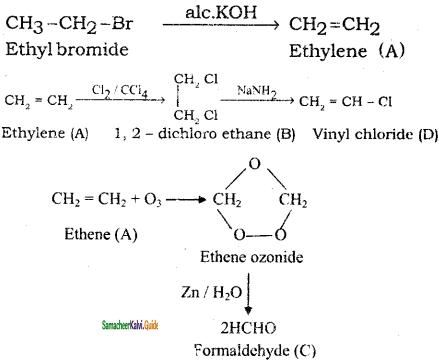
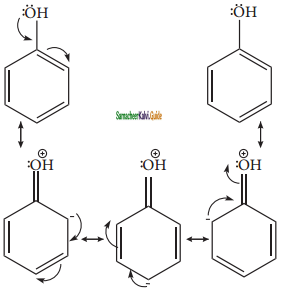
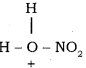 + HSO4–
+ HSO4–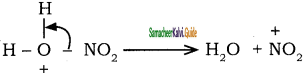
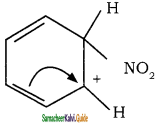
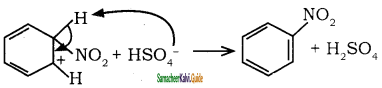
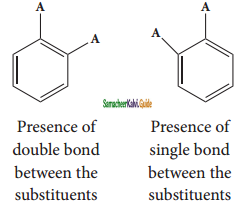
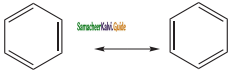
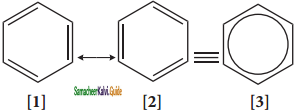
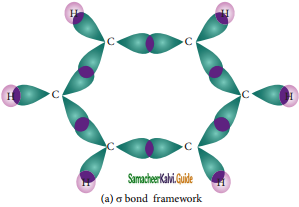
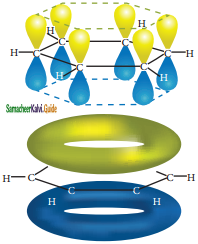
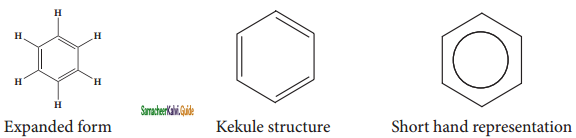
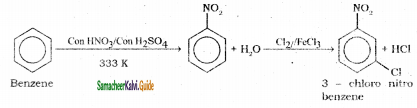
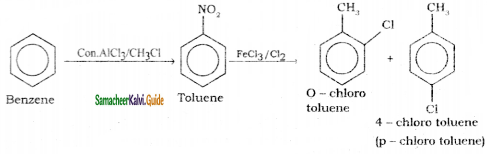
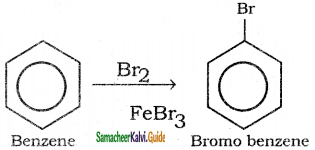
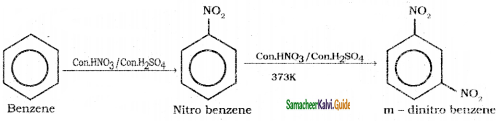
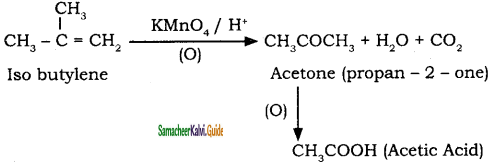
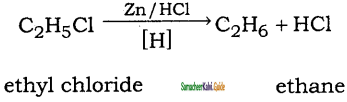
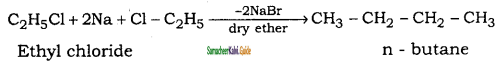
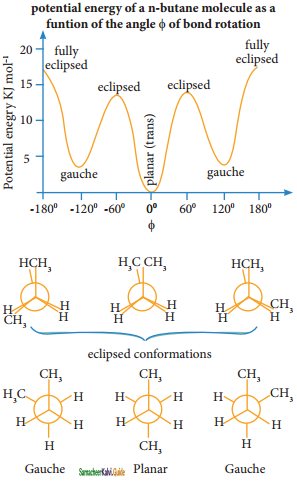
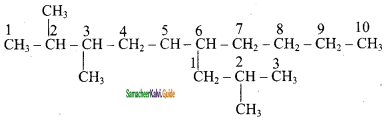
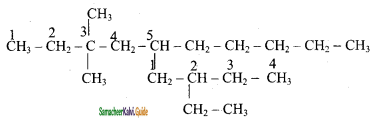
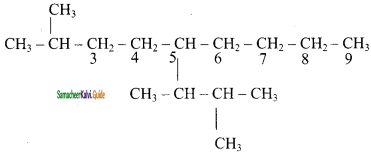
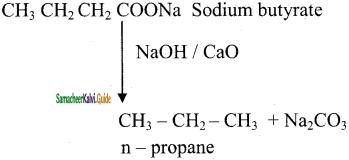
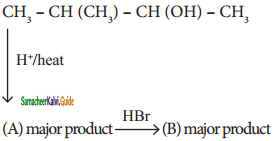
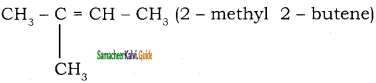
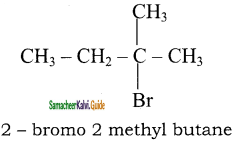
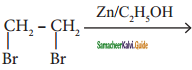
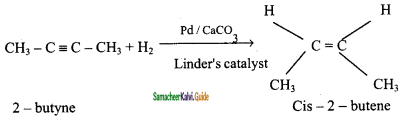
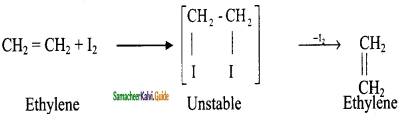
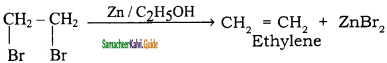
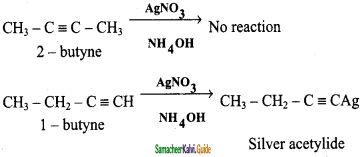
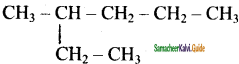
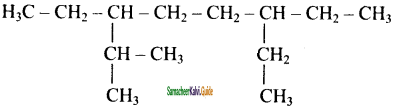
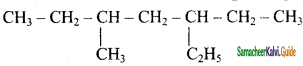
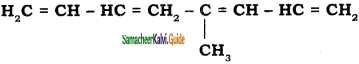
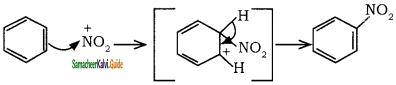
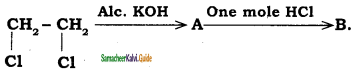 is
is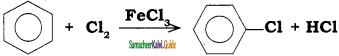

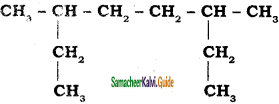
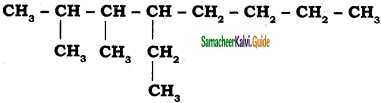
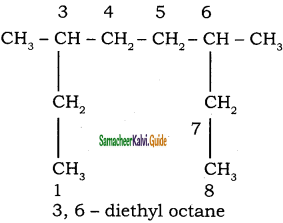
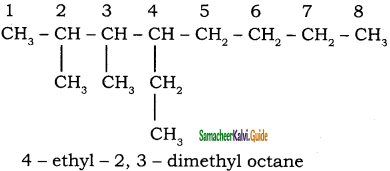
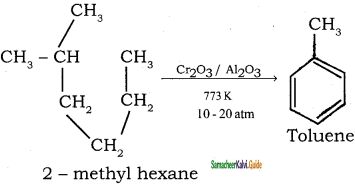
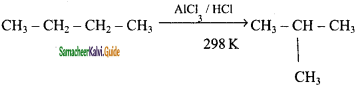
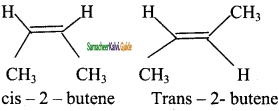
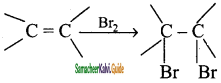
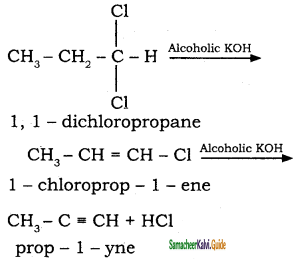
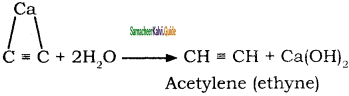
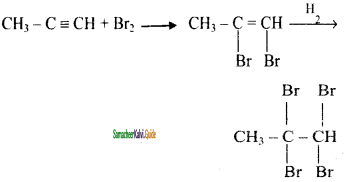
 CH3 – CH3
CH3 – CH3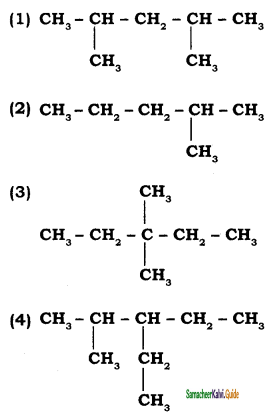
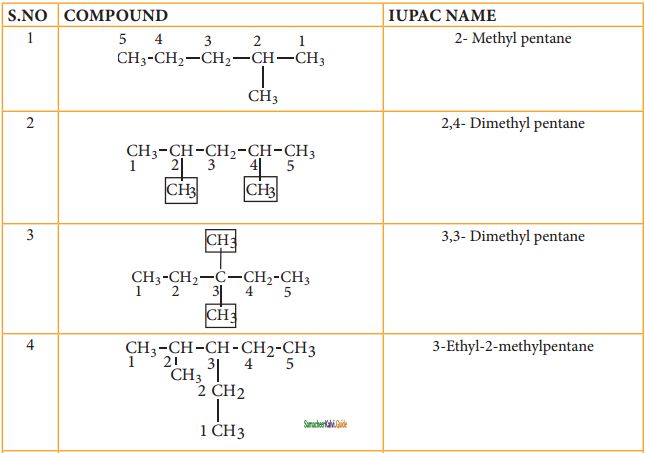
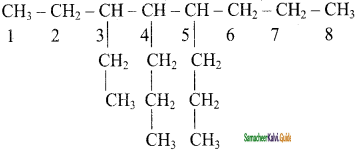
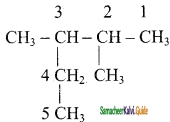
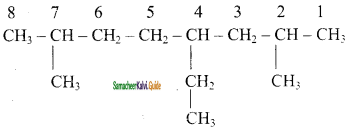
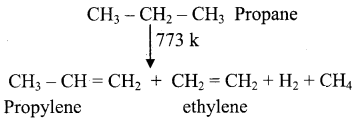
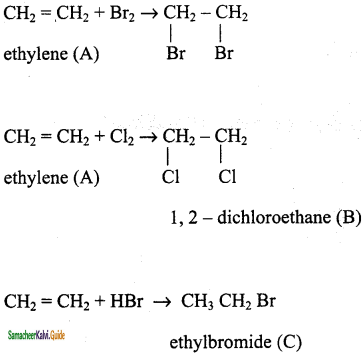
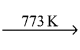 CH2 = CH2 + 2CH4
CH2 = CH2 + 2CH4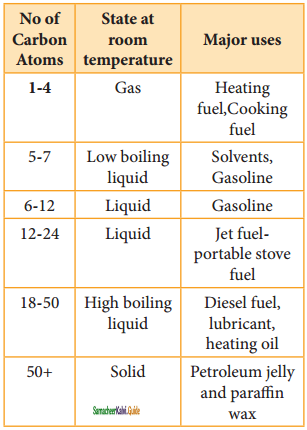
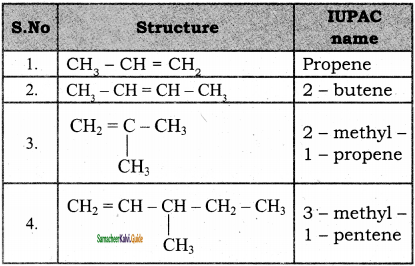
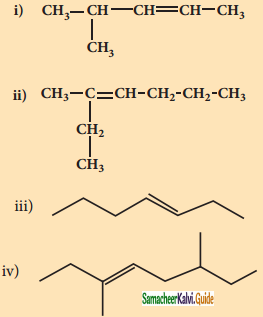
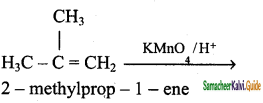
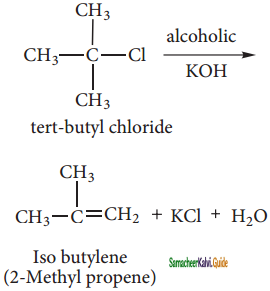
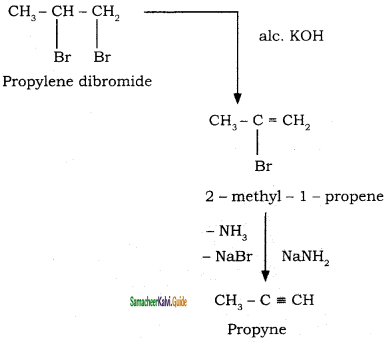
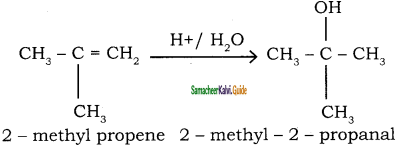
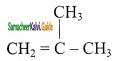 2 – mehyl – 1 – propene
2 – mehyl – 1 – propene