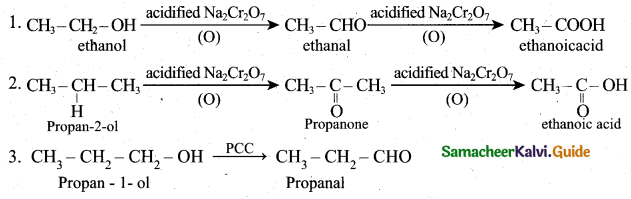
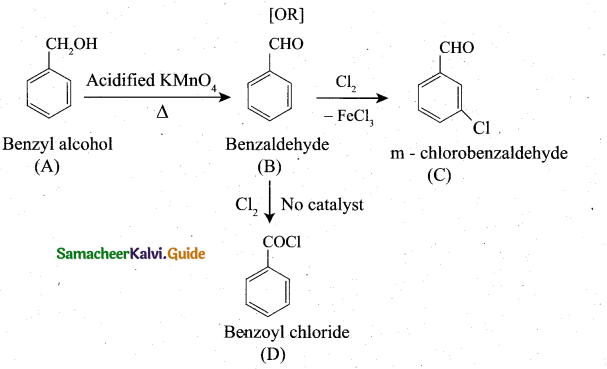
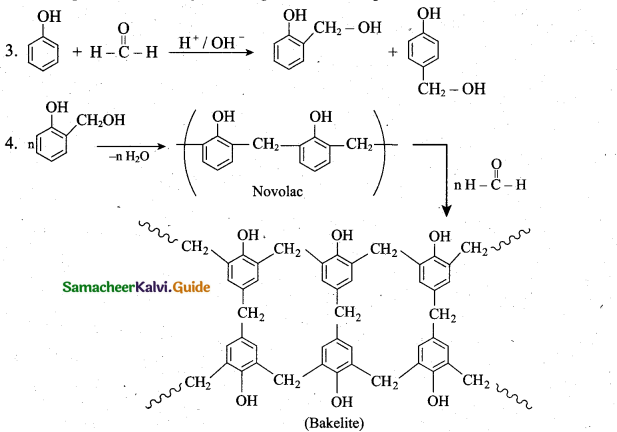
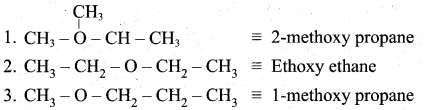Students can download 6th Social Science Term 1 Civics Chapter 2 Achieving Equality Questions and Answers, Notes, Samacheer Kalvi 6th Social Science Guide Pdf helps you to revise the complete Tamilnadu State Board New Syllabus, helps students complete homework assignments and to score high marks in board exams.
Tamilnadu Samacheer Kalvi 6th Social Science Civics Solutions Term 1 Chapter 2 Achieving Equality
Samacheer Kalvi 6th Social Science Achieving Equality Text Book Back Questions and Answers
I. Choose the correct answer
Question 1.
Which one of the following is not the reason for Prejudice?
(a) Socialization
(b) Economic Benefits
(c) Authoritarian personality
(d) Geography
Answer:
(d) Geography
Question 2.
Discrimination done on the basis of gender is referred to as
(a) gender discrimination
(b) caste discrimination
(c) religious discrimination
(d) inequality
Answer:
(a) gender discrimination
Question 3.
Gender-based stereotypes are often portrayed in …………….
(a) films
(b) advertisements
(c) TV serials
(d) All the these
Answer:
(d) All the these
![]()
Question 4.
Name the book/s written by A.P.J. Abdul Kalam
(a) India 2020
(b) Wings of Fire
(c) Ignited Minds
(d) All of these
Answer:
(d) All of these
Question 5.
A.P.J. Abdul Kalam was conferred Bharat Ratna in the year …………….
(a) 1997
(b) 1996
(c) 1995
(d) 1994
Answer:
(a) 1997
Question 6.
Viswanathan Anand became India’s first grandmaster in the year.
(a) 1985
(b) 1986
(c) 1987
(d) 1988
Answer:
(d) 1988
![]()
Question 7.
In which sport Ilavazhagi excels …………….
(a) Chess
(b) Wrestling
(c) Carrom
(d) Tennis
Answer:
(c) Carrom
Question 8.
Which article of the Constitution says discrimination against any citizen on grounds only of religion, race, caste, sex, place of birth or any of them is not permitted?
(a) 14(1)
(b) 15(1)
(c) 16(1)
(d) 17(1)
Answer:
(b) 15(1)
Question 9.
B.R. Ambedkar was conferred Bharat Ratna in the year …………….
(a) 1990
(b) 1989
(c) 1988
(d) 1987
Answer:
(a) 1990
Question 10.
As per the 2011 Census the highest literate district in Tamil Nadu
(a) Namakkal
(b) Salem
(c) Kanyakumari
(d) Sivagangai
Answer:
(c) Kanyakumari
II. Fill in the blanks
- People have false belief and ideas on others is called …………….
- A.P.J. Abdul Kalam was born in …………….
- ……………. was the first recipient of Rajiv Gandhi Khel Ratna award, India’s highest Sport honour.
- ……………. was the independent India’s first Law Minister.
- As per the 2011 census the lowest Sex ratio was in …………….
Answer:
- Prejudice
- 1931
- Viswanathan Anand
- Dr.B.R.Ambedkar
- Dharmapuri
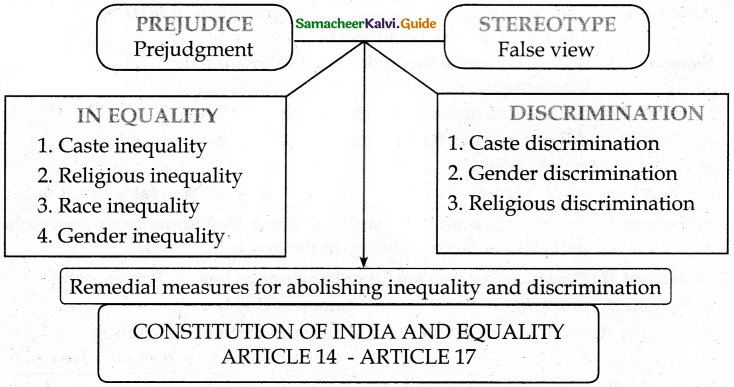
III. Match the following

Answer:
1. – e
2. – d
3. – b
4. – c
5. – a
IV. Answer the following questions
Question 1.
What is Prejudice?
Answer:
- Prejudice means to judge other people in a negative or inferior manner, without knowing much about them.
- It happens when people have false beliefs and ideas.
Question 2.
What is a stereotype?
Answer:
- A stereotype is a false view or idea about something.
- For example, girls are not good at sports.
- When prejudice gets stronger, it develops into a stereotype.
![]()
Question 3.
What is discrimination?
Answer:
- Discrimination can be defined as negative actions towards people.
- Discrimination can happen on the basis of colour, class, religion, and gender, etc.
Question 4.
What are the articles that are discussing equality in the Indian constitution?
Answer:
- Article 14 of the constitution
- Article 15(1) of the constitution
V. Answer the following in detail
Question 1.
What are the causes of prejudice?
Answer:
- Socialization
- Conforming behaviours
- Economic benefits
- Authoritarian personality
- Ethno – centrism
- Group closure
- Conflicts
![]()
Question 2.
Write any two types of discrimination?
Answer:
(a) Gender discrimination:
- Gender discrimination refers to health, education, economic and political inequalities between men and women in India.
- For example a girl is not allowed to go to college after finishing her schooling.
- Most of the girls are not allowed to select a career of their choice, rather they are forced into marriage.
- In some families, girls are not allowed to wear modem dresses while boys in such families often wear modem dresses.
(b) Religious discrimination:
- Religious discrimination is unequal treatment of an individual or group based on their beliefs.
- Religious discrimination has been around for a long time.
- There have been problems between people of different religions for thousands of years.
- Some people are not allowed to enter in public places; especially the places of worship, because they belong to another religion.
- Some religious people often end up in conflict with each other because of their rituals and way of life.
![]()
Question 3.
Explain the solution to the removal of inequality and discrimination in Indian society.
Answer:
The remedial measures for abolishing inequality and discrimination in Indian society are as follows.
- Wider access to quality based services like healthcare and education for all.
- Be aware of current gender bias.
- Make women more visible in public life and institution to eradicate gender disparity.
- Be open to learning about other religions.
- Promoting community dining in the classroom may help the students to sit together without any bias of caste, religion, or gender.
- Socialise with people of all types outside the home.
- Effective implementation of laws.
VI. Projects and Activities
- Split the class into small groups, discuss with your peer group on discrimination and write a report on it.
- Collect information about any two famous personalities who faced prejudice and discrimination.
VII. HOTS
Question 1.
Various discrimination in India. Discuss.
Answer:
- The different forms of inequalities such as caste inequality, religious inequality, race inequality, or gender inequality give rise to discrimination.
- Discrimination can happen on the basis of colour, class, religion gender, etc.
- Treating dark-skinned people differently from fair-skinned people, giving more importance to people of higher than to those of lower caste, and thinking boys are smarter than girls are all thoughts of discrimination.
- Article 15(1) of the constitution states that the State not to discriminate against any citizen on grounds only of religion, race, caste, sex, place of birth, or any of them.
VIII. Life Skill
- How can you fight against prejudice and discrimination in your village?
Samacheer Kalvi 6th Social Science Achieving Equality Additional Important Questions and Answers
I. Choose the correct answer
Question 1.
Apartheid was practiced in ……………
(a) Germany
(b) Australia
(c) South Africa
(d) India
Answer:
(c) South Africa
Question 2.
Viswanathan Anand was born in _______
(a) Chennai
(b) Bengaluru
(c) Thiruvananthapuram
(b) Vishakapatnam
Answer:
(a) Chennai
![]()
Question 3.
The chairman of the Drafting Committee of India’s constituent Assembly was ……………
(a) Nehru
(b) Dr. Ambedkar
(c) Mount Batten
(d) Dr. Abdul kalam
Answer:
(b) Dr. Ambedkar
Question 4.
Apartheid, the racist policy was brought to an end by _______
(a) Martin Luther
(b) Nelson Mandela
(c) Mother Theresa
(d) Tagore
Answer:
(b) Nelson Mandela
Question 5.
Mr. Viswanathan Anand won the World Junior Championship in ……………
(a) Chess
(b) Carrom
(c) Cricket
(d) High jump
Answer:
(a) Chess
II. Answer the following questions
Question 1.
Mention the types of prejudice
Answer:
- Gender prejudice
- Racial prejudice
- Class prejudice
- Disability prejudice
Question 2.
Name the books written by Dr. APJ Abdul Kaiam.
Answer:
Dr. APJ Abdul Kalam wrote many books. The famous among them are –
- India 2020
- Wings of Fire
- Ignited Minds
- The Luminous Sparks
- Mission India.
Question 3.
What is meant by constitution?
Answer:
A constitution is a set of rules and regulations guiding the administration of a country.
![]()
Question 4.
What does Article 15(1) of our constitution state about discrimination?
Answer:
The state shall not discriminate against any citizen on grounds only of religion, race, caste, sex and place of birth or any of them.
Question 5.
What does Article 14 of our constitution state about equality?
Answer:
Article 14 provides equality before law or equal protection within the territory of India and prohibits unreasonable discrimination between persons.
III. Mind map




















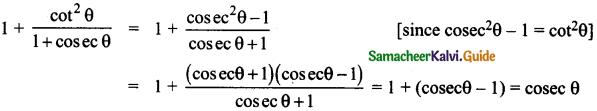
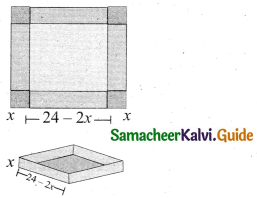
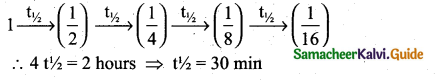
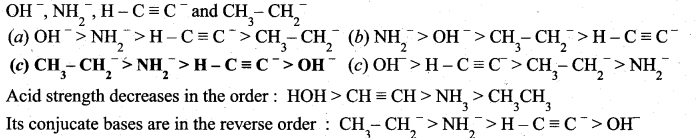

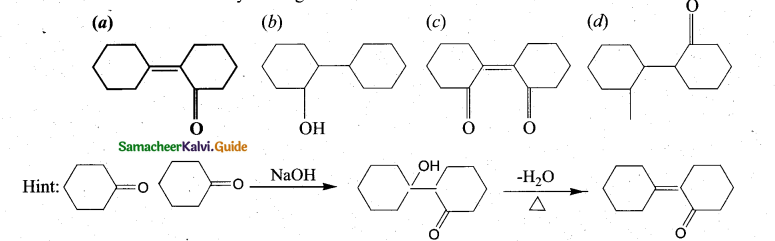
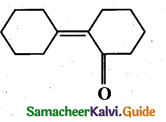

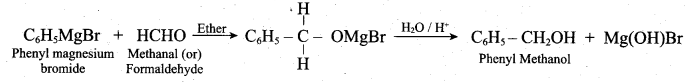
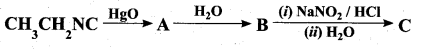
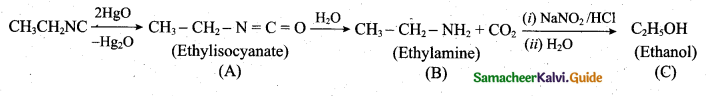
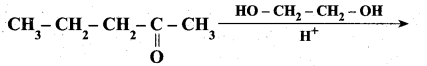
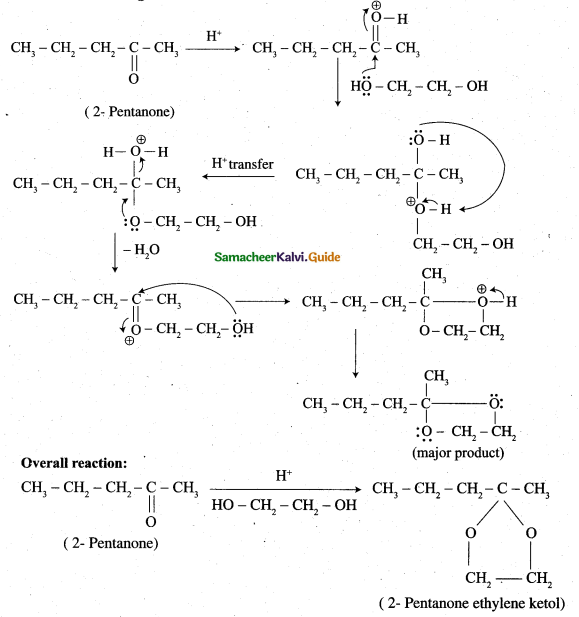
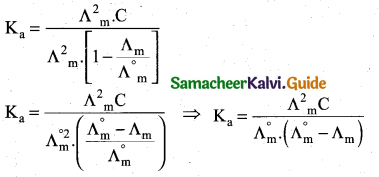
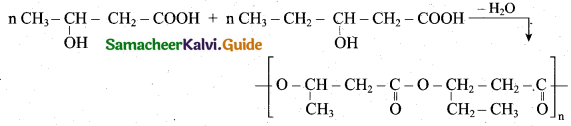
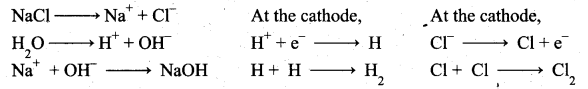
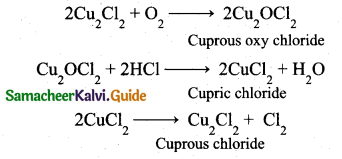
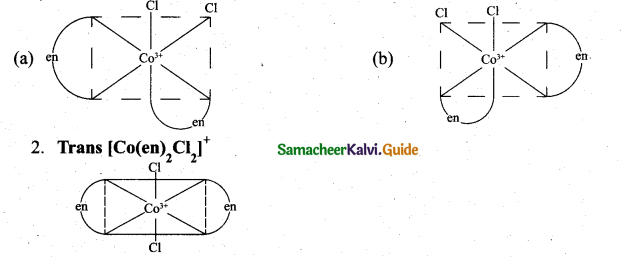



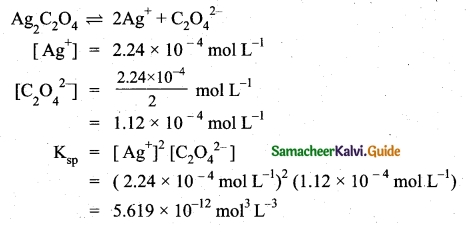
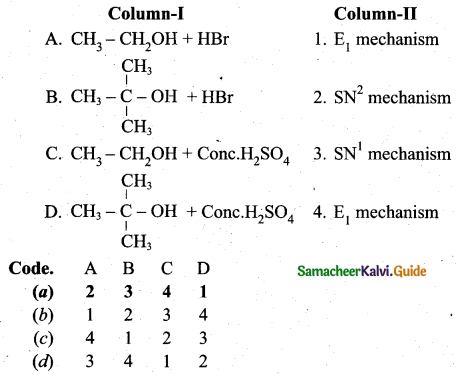
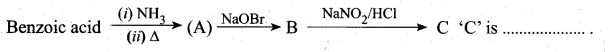
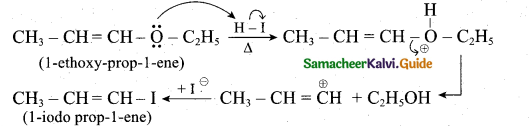
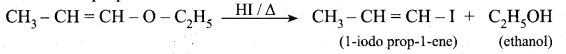

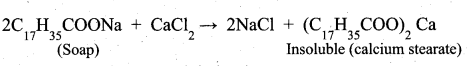
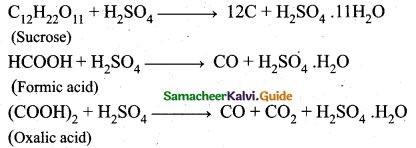

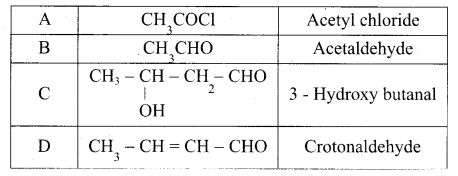
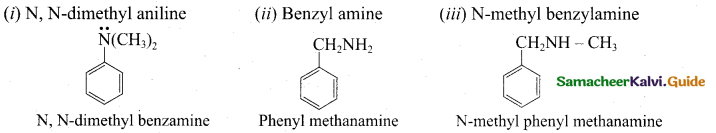
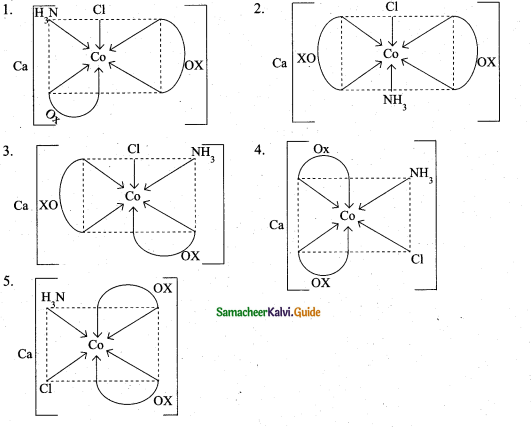
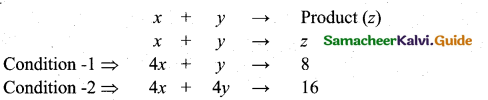 z = k [x]m [y]n …… (1)
z = k [x]m [y]n …… (1)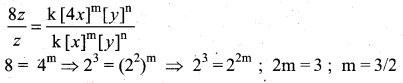
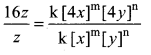
 Answer:
Answer:


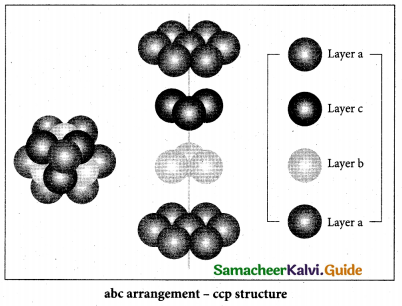

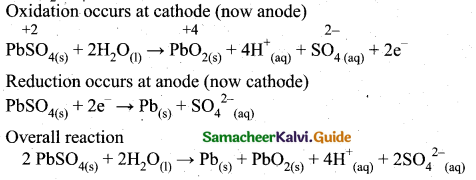
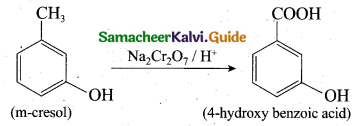
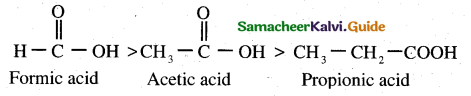

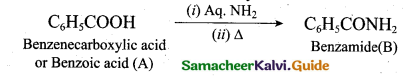
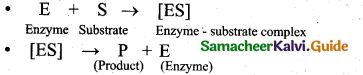











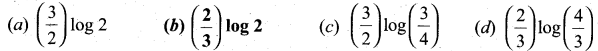


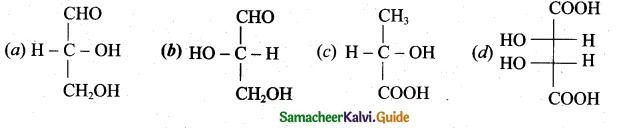
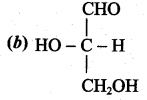
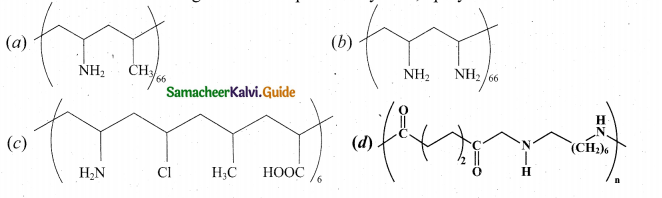
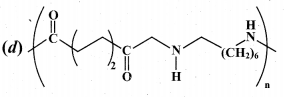
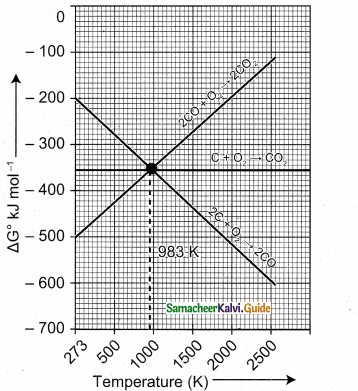
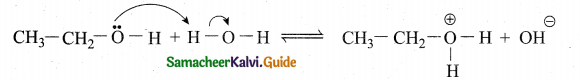
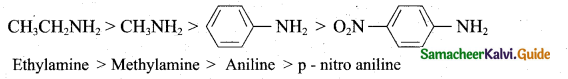
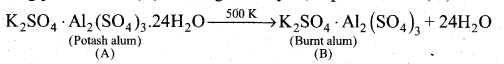

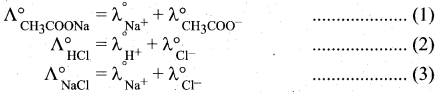
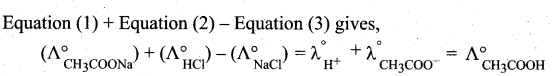
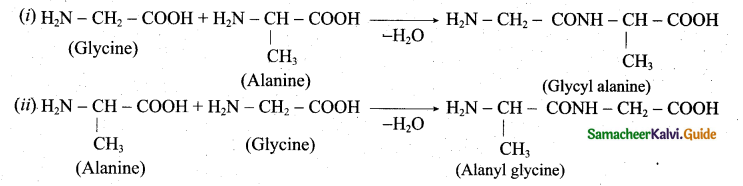
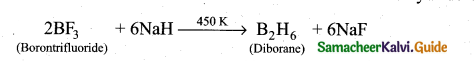

 (2)
(2)