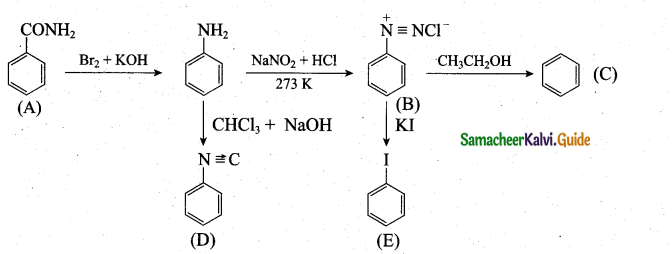
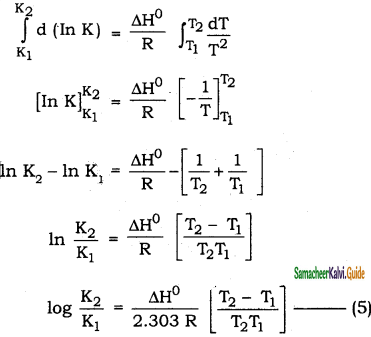
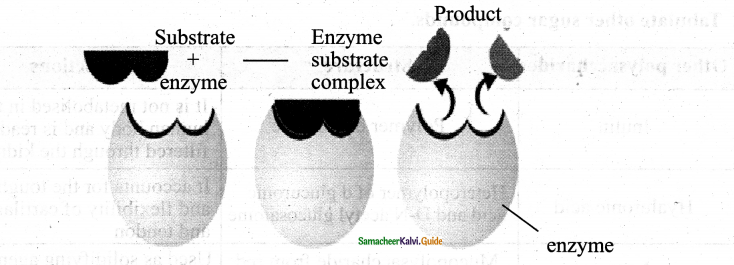
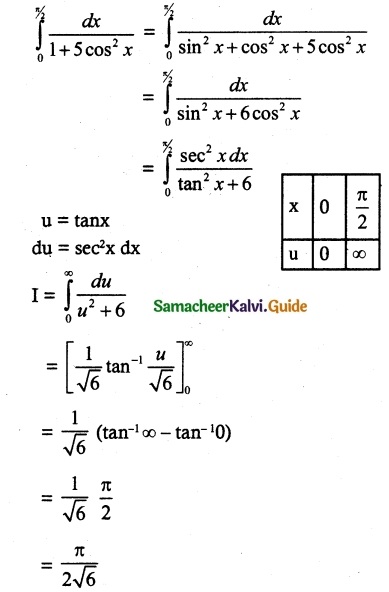
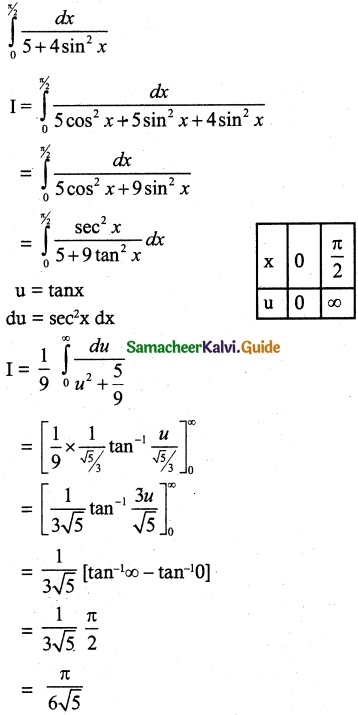
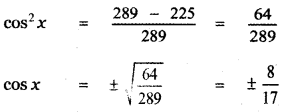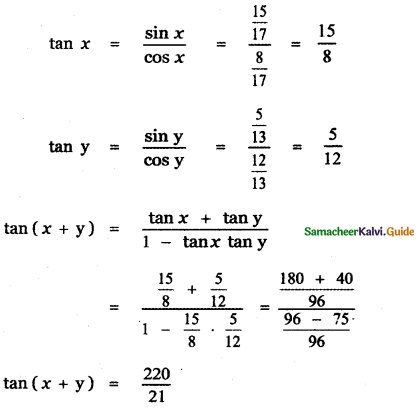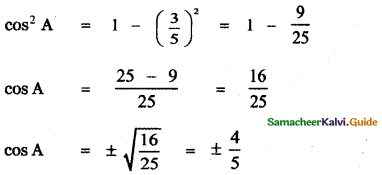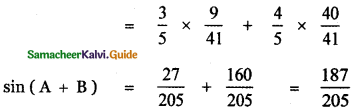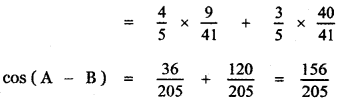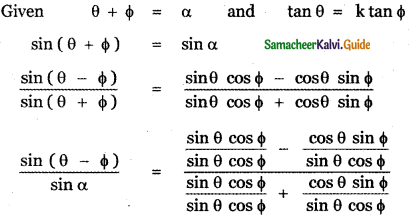Students can Download 10th Tamil Chapter 6.7 திருக்குறள் Questions and Answers, Summary, Notes, Samacheer Kalvi 10th Tamil Guide Pdf helps you to revise the complete Tamilnadu State Board New Syllabus, helps students complete homework assignments and to score high marks in board exams.
Tamilnadu Samacheer Kalvi 10th Tamil Solutions Chapter 6.7 திருக்குறள்
கற்பவை கற்றபின்
![]()
Question 1.
புதுக்கவிதையையும் குறளையும் ஒப்பிட்டுப் பேசுக.
புதுக்கவிதை
தக்காளியையும் வெண்டைக்காயையும்
தள்ளுவண்டிக்காரர் தராசில் நிறுக்கையில்,
தள்ளி நிற்கும் பிள்ளை
அவசியமாகக் கேட்கும் ஆயிரம் ரூபாயை
எப்படிக் கொடுக்க என்றே அவர் மனம் யோசிக்கும்….
“அத்தனைக் காய்களையும் விற்றால்தான்
மீதி ஐந்நூறாவது மிஞ்சும்; என்ன செய்ய…”
காய்கறி வாங்கியவர்
கவனக் குறைவாகக் கொடுத்த
இரண்டாயிரம் ரூபாயைக்
கூப்பிட்டுத் தந்துவிட்டுப்
பிள்ளைக்கு உதவ யாரிடம் கேட்கலாம்.
என்பதை அடுத்தபடி யோசிக்கும் அவர் மனம்!
![]()
குறள்
அருளொடும் அன்பொடும் வாராப் பொருளாக்கம்
புல்லார் புரள விடல்
Answer:
மற்றவர்களிடம் இரக்கமும் அன்பும் இல்லாமல் ஈட்டும் பொருளை ஏற்றுக் கொள்ளாது நீக்கிவிடவேண்டும் என்ற கொள்கையைப் பின்பற்றிய தள்ளுவண்டிக்காரர், காய்கறி வாங்கியவர் கவனக் குறைவாகக் கொடுத்த இரண்டாயிரம் ரூபாயைத் தான் வைத்துக்கொள்ளாமல் காய்கறி வாங்கியவரை கூப்பிட்டுக் கொடுத்துவிட்டார்.
தனக்குத் தேவை மிகுதியாய் இருப்பினும் தள்ளுவண்டிக்காரர் அறவழியில் செல்வம் ஈட்டுவதையே விரும்புகிற ஒரு நல்ல பண்பாளர்.
Question 2.
குறிப்புகள் உணர்த்தும் குறளின் கருத்துகள் குறித்துக் கலந்துரையாடுக.
அ) அவருக்கு அறிவும் அதிகம்; படிப்பும் அதிகம். யாராலும் அவருக்கெதிராகச் செயல்பட முடியாது.
Answer:
பாலா : அவருக்கு அறிவும் அதிகம்; படிப்பும் அதிகம். யாராலும் அவருக்கெதிராகச் செயல்பட முடியாது.
மாலா : குடிசெயல்வகை எனும் அதிகாரத்தில் 1022 வது குறளில் விடாமுயற்சி, சிறந்த
அறிவாற்றல் உடையவனின் குடி உயர்ந்து விளங்கும்.
ஆ) எச்சரிக்கையாக இரு. பழகுபவர்களில் யார் நல்லவர், யார் கெட்டவர் என்று கண்டுபிடிப்பது கடினம்.
Answer:
பாலா : எச்சரிக்கையாக இரு. பழகுபவர்களில் யார் நல்லவர், யார் கெட்டவர் என்று
கண்டுபிடிப்பது கடினம்.
மாலா : கூடாநட்பு எனும் அதிகாரத்தில் 828-ஆவது குறளில் பகைவர் நம்மை வணங்கி தொழும்போது கையில் கத்தியை மறைத்து வைத்திருப்பர்.
இ) அவர் குடும்பம் முன்னேறியிருப்பதற்குக் காரணம் தொடர்ந்து முயல்வதும் தொழிலில் அவருக்கிருந்த அறிவும்.
பாலா : அவர் குடும்பம் முன்னேறியிருப்பதற்குக் காரணம் தொடர்ந்து முயல்வதும் தொழிலில் அவருக்கிருந்த அறிவும்.
மாலா : ஊக்கமுடைமை எனும் அதிகாரத்தில் 594-ஆவது குறளில் தளராது ஊக்கத்தோடு உழைப்பவனிடம் தொடர்ந்து செல்வம் சேரும்.
![]()
ஈ) வாழ்க்கையில் நன்றாகப் பொருளீட்டு. அதுதான் உன்னைத் தாழ்வாக நினைத்தவர்களின் வாயை அடைக்கும்.
Answer:
பாலா : வாழ்க்கையில் நன்றாகப் பொருளீட்டு. அதுதான் உன்னைத் தாழ்வாக
நினைத்தவர்களின் வாயை அடைக்கும்.
மாலா : பொருள் செயல்வகை எனும் அதிகாரத்தில் 759-ஆவது குறளில் பொருள் இல்லாரை எல்லாரும் இகழ்வர். பொருள் உள்ளவரை போற்றுவர்.
உ) அக்கம்பக்கத்தாரிடம் பழகாமல் ஒதுங்கியே இருந்தார்; துணைக்கும் யாரும் இல்லை. இப்போது உடம்பு சரியில்லாதபோது யாருமின்றித் திண்டாடுகிறார்.
Answer:
பாலா : அக்கம்பக்கத்தாரிடம் பழகாமல் ஒதுங்கியே இருந்தார்; துணைக்கும் யாரும் இல்லை.
இப்போது உடம்பு சரியில்லாதபோது யாருமின்றித் திண்டாடுகிறார்.
மாலா : அன்புடைமை எனும் அதிகாரத்தில் 80-ஆவதுகுறளில் பிறரிடம் அன்போடு வாழ்பவன் உயிருடன் கூடிய உடல். அன்பு இல்லாததால் உயிரற்ற எலும்புக்கூட்டுக்குச் சமம்.
குறுவினா
Question 1.
கரப்பிடும்பை இல்லார் – இத்தொடரின் பொருள் கூறுக.
Answer:
தம்மிடமுள்ள பொருளை மறைத்து வைத்தல் என்னும் துன்பம் தராத நல்லவர்.
Question 2.
தஞ்சம் எளியன் பகைக்கு – இவ்வடிக்குரிய அசைகளையும் வாய்பாடுகளையும் எழுதுக.
Answer:

![]()
Question 3.
வறுமையின் காரணமாக உதவி கேட்டு வருபவரின் தன்மானத்தை எள்ளிநகையாடுவது குறித்துக் குறளின் கருத்து என்ன?
Answer:
இகழ்ந்து ஏளனம் செய்யாமல் பொருள் கொடுப்பவரைக் கண்டால் இரப்பவரின் உள்ளத்தின் உள்ளே மகிழ்ச்சி பொங்கும்.
Question 4.
பின்வருவனவற்றுள் கூரான ஆயுதம் எது என்று செந்நாப்போதார் கூறுகிறார்? ஏன் என்பதை எழுதுக.
Answer:
பெரிய கத்தி, இரும்பு ஈட்டி, உழைத்ததால் கிடைத்த ஊதியம், வில்லும் அம்பும்
- கூரான ஆயுதம்: உழைத்ததால் கிடைத்த ஊதியம்.
- காரணம்: இதுவே அவனுடைய பகைவனை வெல்லும் கூரான ஆயுதம்.
![]()
சிறுவினா
Question 1.
வள்ளுவம், சிறந்த அமைச்சருக்குக் கூறிய இலக்கணங்கள் நமக்கும் பொருந்துவதைக் குறள்வழி விளக்குக.
Answer:

தொழில் செய்வதற்குத் தேவையான கருவி, அதற்கு ஏற்ற காலம், செயலின் தன்மை, செய்யும் முறை ஆகியவற்றை அறிந்து அரிய செயல்களைச் செய்தல் வேண்டும்.

மனவலிமை, குடிகளைக் காத்தல், ஆட்சிமுறைகளைக் கற்றல், நூல்களைக் கற்றல், விடாமுயற்சி ஆகிய ஐந்தும் சிறப்பாக அமைய வேண்டும்.
சூழ்ச்சிகள்:

இயற்கையான நுண்ணறிவு, நூலறிவு இவற்றை உடையவர்கள் முன் எந்த சூழ்ச்சியும் நிற்க இயலாது.
நடைமுறைகளை எறிதல்:

ஒரு செயலைச் செய்வதற்குரிய முறைகளை நூல்வழியாக அறிந்திருப்பினும் உலகியல் நடைமுறைகளை அறிந்து செயல்பட வேண்டும்.
![]()
Question 2.
பலரிடம் உதவி பெற்றுக் கடின உழைப்பால் முன்னேறிய ஒருவர், அவருக்கு உதவிய நல்ல உள்ளங்களையும் சுற்றங்களையும் அருகில் சேர்க்கவில்லை. அவருக்கு உணர்த்தும் நோக்கில் வள்ளுவர் குறிப்பிடும் கருத்துகள் யாவை?
Answer:
பகைவரின் வலிமை:

சுற்றத்தாரிடம் ஒருவர் அன்பு இல்லாமலும் பொருந்திய துணை இல்லாமலும் வலிமை இல்லாமலும் இருந்தால் அவரால் பகைவரின் வலிமையை எதிர்கொள்ள முடியாது.
பகைக்கு ஆட்படல்:

மனதில் துணிவு இல்லாதவராய் அறிய வேண்டியவற்றை அறியாதவராய் பொருந்தும் பண்பு இல்லாதவராய், பிறருக்குக் கொடுத்து உதவாதவராய் இருந்தால் எளிதில் பகைக்கு ஆட்பட நேரும்.
கூடுதல் வினாக்கள்
இலக்கணக் குறிப்பு.
கருவியும் காலமும் – எண்ணும்மை
அருவினை – பண்புத்தொகை
வன்கண் – பண்புத்தொகை
வந்த பொருள் – பெயரெச்சம்
வாராப்பொருளாக்கம் – ஈறுகெட்ட எதிர்மறைப் பெயரெச்சம்
செய்க – வியங்கோள் வினைமுற்று
நீள்வினை – வினைத்தொகை
![]()
பகுபத உறுப்பிலக்கணம்.

பலவுள் தெரிக
Question 1.
பொருளல் லவரைப் பொருளாகச் செய்யும்
பொருளல்ல தில்லை பொருள் – இக்குறளில் பயின்று வரும் அணி.
அ) உவமையணி
ஆ) பொருள் பின்வருநிலையணி
இ) சொல்பின்வருநிலை அணி
ஈ) சொற்பொருள் பின் வருநிலையணி
Answer:
ஈ) சொற்பொருள் பின் வருநிலையணி
Question 2.
குன்றேறி யானைப்போர் கண்டற்றால் தன்கைத்தொன்
றுண்டாகச் செய்வான் வினை – இக்குறளில் பயின்று வரும் அணி.
அ) உவமையணி
ஆ) உருவக அணி
இ) வேற்றுமை அணி
ஈ) பிறிது மொழிதல் அணி
Answer:
அ) உவமையணி
Question 3.
இன்மையின் இன்னாத தியாதெனின் இன்மையின்
இன்மையே இன்னா தது – இக்குறளில் பயின்று வரும் அணி.
அ) சொல்பின்வருநிலையணி
ஆ) சொற்பொருள் பின்வருநிலையணி
இ) உவமையணி
ஈ) உருவக அணி
Answer:
ஆ) சொற்பொருள் பின்வருநிலையணி
![]()
Question 4.
மக்களே போல்வர் கயவர்; அவரன்ன
ஒப்பாரி யாம்கண்ட தில். – இக்குறளில் பயின்று வரும் அணி.
அ) உவமை அணி
ஆ) உருவக அணி
இ) வேற்றுமை அணி
ஈ) பிறிதுமொழிதல் அணி
Answer:
அ) உவமை அணி
Question 5.
தேவர் அனையர் கயவர் அவரும்தாம்
மேவன செய்தொழு கலான் – இக்குறளில் பயின்று வரும் அணி.
அ) தற்குறிப்பேற்ற அணி
ஆ) வஞ்சப்புகழ்ச்சி அணி
இ) உவமை அணி
ஈ) உருவக அணி
Answer:
ஆ) வஞ்சப்புகழ்ச்சி அணி
Question 6.
சொல்லப் பயன்படுவர் சான்றோர்; கரும்புபோல்
கொல்லப் பயன்படும் கீழ் – இக்குறளில் பயின்று வரும் அணி.
அ) உவமையணி
ஆ) உருவக அணி
இ) பிறிதுமொழிதல் அணி
ஈ) வேற்றுமை அணி
Answer:
அ) உவமையணி
![]()
Question 7.
சிறந்த அமைச்சருக்குரிய குண நலன்கள் ………………
அ) 4
ஆ) 5
இ) 3
ஈ) 6
Answer:
ஆ) 51
Question 8.
விடாமுயற்சி, சிறந்த அறிவாற்றல் இவ்விரண்டையும் இடைவிடாமல் பின்பற்றுபவரின் குடி………………
அ) உயர்ந்து விளங்கும்
ஆ) தாழ்ந்து நிற்கும்
இ) வாடிப் போகும்
ஈ) காணாமல் நீங்கும்
Answer:
அ) உயர்ந்து விளங்கும்
குறுவினா
Question 1.
எத்தகைய அரிய செயலைச் செய்பவர் அமைச்சர் ஆவார்?
Answer:
தொழில் செய்வதற்குத் தேவையான கருவி, அதற்கு ஏற்ற காலம், செயலின் தன்மை, செய்யும் முறை ஆகியவற்றை அறிந்து அரிய செயலைச் செய்பவர் அமைச்சர் ஆவார்.
Question 2.
அமைச்சருக்குரிய ஐந்து சிறப்புகள் யாவை?
Answer:
மனவலிமை, குடிகளைக் காத்தல், விடா முயற்சி, ஆட்சி முறைகளைக் கற்றல், நூல்களைக் கற்றல்.
![]()
Question 3.
எத்தகைய அமைச்சர்களுக்கு முன் சூழ்ச்சியும் நிற்க இயலாது?
Answer:
இயற்கையான நுண்ணறிவு, நூலறிவு இவற்றையுடைய அமைச்சர்களுக்கு முன் சூழ்ச்சிகள் நிற்க இயலாது.
Question 4.
ஓர் அமைச்சன் எவற்றை அறிந்து செயல்பட வேண்டும்?
Answer:
ஒரு செயலைச் செய்வதற்குரிய முறைகளை நூல் வழியாக அறிந்திருப்பினும் உலகியல் நடைமுறைகளை அறிந்து செயல்பட வேண்டும்.
Question 5.
அறத்தையும் இன்பத்தையும் தருவது எது?
Answer:
முறையறிந்து தீமையற்ற வழியில் சேர்ந்த பொருள் ஒருவனுக்கு அறத்தையும் தரும் இன்பத்தையும் தரும்.
Question 6.
எத்தகையப் பொருளை ஏற்காமல் நீக்கிவிட வேண்டும்?
Answer:
மற்றவர்களிடம் இரக்கமும் அன்பும் இல்லாமல் ஈட்டும் பொருளை ஏற்றுக்கொள்ளாமல் நீக்கிவிட வேண்டும்.
![]()
Question 7.
பொருளல்லவரைப் பொருளாகச் செய்வது எது? ஏன்?
Answer:
- ஒரு பொருட்டாக மதிக்கத்தகாதவரையும் பிறர் மதிக்கும்படி செய்வது செல்வம்.
- ஏனெனில் அதைவிட சிறந்த பொருள் உலகில் வேறு எதுவும் இல்லை.
Question 8.
ஒருவன் தன் கைப்பொருளைக் கொண்டு செய்யும் செயல் எதற்கு உவமையாகக் கூறப்பட்டுள்ளது?
Answer:
ஒருவன் தன் கைப்பொருளைக் கொண்டு செய்யும் செயலானது, குன்றின் மேல் பாதுகாப்பாக நின்று கொண்டு யானைப் போரைக் காண்பது போன்றது.
![]()
Question 9.
கூடா நட்பு குறித்து வள்ளுவர் கூறும் செய்தி யாது?
Answer:
கொலைக் கருவி:
பகைவரின் தொழுது நிற்கும் கையின் உள்ளேயும் கொலைக் கருவி மறைந்து இருக்கும்.
வஞ்சகம்:
பகைவரின் அழுத கண்ணீர் உள்ளும் வஞ்சகம் மறைந்து இருக்கும்.
Question 10.
யாரால் பகைவரின் வலிமையை எதிர்கொள்ள முடியாது?
Answer:
- சுற்றத்தாரிடம் அன்பு இன்மை
- பொருந்திய துணை இன்மை
- வலிமையின்மை
இவற்றையுடையவன் பகைவரின் வலிமையை எதிர்கொள்ள முடியாது.
Question 11.
ஒருவன் எளிதில் பகைக்கு ஆட்பட நேரிடுவது எப்போது?
Answer:
- மனதில் துணிவு இல்லாமை
- அறியவேண்டியவற்றை அறியாமை
- பொருந்தும் பண்பு இல்லாமை
- பிறருக்குக் கொடுத்து உதவாமை
மேற்கண்ட செயல்களை உடையவர் எளிதில் பகைக்கு ஆட்பட நேரும்.
![]()
Question 12.
ஒருவனது குடி எப்போது சிறந்து விளங்கும்?
Answer:
விடாமுயற்சி, சிறந்த அறிவாற்றல் இவ்விரண்டையும் இடைவிடாமல் பின்பற்றுபவரின் குடி சிறந்து விளங்கும்.
Question 13.
உலகத்தார் யாரை உறவாகக் கொண்டு போற்றுவார்?
Answer:
குற்றம் இல்லாமல் தன் குடிப்பெருமையை உயரச் செய்து வாழ்பவரை உலகத்தார் உறவாகக் கொண்டு போற்றுவர்.
Question 14.
வறுமையின் கொடுமை முழுவதும் கெடும் எப்போது?
Answer:
தம்மிடமுள்ள பொருளை மறைத்து வைத்தல் என்னும் துன்பம் தராத நல்லாரைக் கண்டால் வறுமையின் கொடுமை முழுவதும் கெடும்.
![]()
Question 15.
யாருடைய உள்ளத்தில் மகிழ்ச்சிப் பொங்கும். எப்போது?
Answer:
இகழ்ந்து ஏளனம் செய்யாமல் பொருள் கொடுப்பவரைக் கண்டால் இரப்பவரின் உள்ளத்தில் மகிழ்ச்சி பொங்கும்.
Question 16.
மக்கள், கயவர் குறித்து வள்ளுவர் கூறிய செய்தி யாது?
Answer:
கயவர் மக்களைப் போலவே இருப்பர், கயவர்க்கும் மக்களுக்கும் உள்ள ஒப்புமை தோற்றம் மட்டுமே வேறெந்த ஒப்புமையும் கிடையாது.
Question 17.
தேவர் கயவர் குறித்து வள்ளுவர் கூறியது யாது?
Answer:
தேவரும் கயவரும் ஒரே தன்மையர், தேவர்களைப் போலவே கயவர்களும் தாம் விரும்புவனவற்றைச் செய்து வாழ்வர்.
![]()
Question 18.
சான்றோர் கயவர் குறித்து வள்ளுவர் கூறுவது யாது?
Answer:
- ஒருவர் தம் குறையை சொல்வதைக் கேட்டவுடன் உதவி செய்பவர் சான்றோர்.
- கரும்பைப் பிழிவது போல நெருக்கிப் பிழிந்தால்தான் பயன்படுபவர் கயவர்.
Question 19.
ஒரு செயலைச் செய்வதற்கு உலகியல் நடைமுறைகளும் அவசியம் என்பதை வலியுறுத்தும் திருக்குறளை எழுதுக.
Answer:
செயற்கை அறிந்தக் கடைத்தும் உலகத்
தியற்கை அறிந்து செயல்.
![]()
Question 20.
ஒரு பொருளாக மதிக்கத்தகாதவரையும் மதிப்புடையவராகச் செய்வது செல்வம் என்பதை எடுத்துரைக்கும் திருக்குறளை எழுதுக.
Answer:
பொருளல் லவரைப் பொருளாகச் செய்யும்
பொருளல்ல தில்லை பொருள்.
Question 21.
மலைமேல் நின்றுகொண்டு யானைப் போரைக் காண்பதனை உவமையாகக் குறிப்பிடும் திருக்குறளை எழுதுக.
Answer:
குன்றேறி யானைப்போர் கண்டற்றால் தன்கைத்தொன்
றுண்டாகச் செய்வான் வினை.
![]()
Question 22.
குற்றமற்றுக் குடிப்பெருமையுடன் வாழ்பவரை உலகத்தார் போற்றுவர் என்பதற்குச் சான்றாக விளங்கும் திருக்குறளை எழுதுக.
Answer:
குற்றம் இலனாய்க் குடிசெய்து வாழ்வானைச்
சுற்றமாச் சுற்றும் உலகு.
Question 23.
வறுமையைப் போன்று துன்பம் தருவது வறுமையே என்று குறிப்பிடும் குறட்பாவினை எழுதுக.
Answer:
இன்மையின் இன்னாத தியாதெனின் இன்மையின் இன்மையே இன்னா தது.
![]()
சிறுவினா
Question 1.
‘குடிச்செயல் வகை என்னும் அதிகாரத்தில் குடி உயர்வு குறித்து வள்ளுவர் கூறும் கருத்து யாது?
Answer:
இடைவிடாமல் பின்பற்றுதல் :
விடா முயற்சி, சிறந்த அறிவாற்றல் இவ்விரண்டையும் இடைவிடாமல் பின்பற்றுபவரின் குடி உயர்ந்து விளங்கும்.
குற்றம் இன்மை :
குற்றம் இல்லாமல் தன் குடிப்பெருமையை உயரச் செய்து வாழ்பவரை உலகத்தார் உறவாகக் கொண்டு போற்றுவர்.
Question 2.
‘கயமை’ என்னும் அதிகாரத்தில் கயவர் குறித்து வள்ளுவர் குறிப்பிடும் செய்தி யாது?
Answer:
தோற்ற ஒப்புமை: கயவருக்கும் மக்களுக்கும் உள்ள ஒப்புமை தோற்றம் மட்டுமே. வேறெதிலும் ஒப்புமை இல்லை.
தேவரும் கயவரும்: தேவரும் கயவரும் ஒரே தன்மையர், தேவர்களைப் போலவே கயவர்களும் தீயவற்றைச் செய்து ஒழுகுவார்கள்
சான்றோர்-கயவர்: ஒருவர் தம் குறையைச் சொல்வதைக் கேட்ட உடனேயே உதவி செய்பவர் சான்றோர். கரும்பைப் பிழிவது போல நெருக்கிப் பிழிந்தால்தான் பயன்படுவர் கயவர்.
![]()
Question 3.
பொருளல் லவரைப் பொருளாகச் செய்யும்
பொருளல்ல தில்லை பொருள் – இப்பாடலில் பயின்று வந்துள்ள அணியைக் கூறி விளக்குக.
Answer:
இக்குறட்பாவில் பயின்று வந்துள்ள அணி சொல் பின்வருநிலையணி.
அணி இலக்கணம்: ஒரு செய்யுளில் முன்னர் வந்த சொல் மீண்டும் மீண்டும் வந்து வெவ்வேறு பொருளைத் தருவது சொல் பின்வருநிலையணி ஆகும்.
விளக்கம் :
இக்குறட்பாவில் பொருள் என்ற சொல் மீண்டும் மீண்டும் வந்து பொருட்டு, செல்வம் ஆகிய வெவ்வேறு பொருளைத் தருகிறது. எனவே, இக்குறட்பாசொல் பின்வருநிலையணிக்குச் சான்றாகிறது.
![]()
Question 4.
குன்றேறி யானைப்போர் கண்டற்றால் தன்கைத்தொன்
றுண்டாகச் செய்வான் வினை – இக்குறட்பாவில் பயின்று வந்துள்ள அணியைக் கூறி விளக்குக.
Answer:
இக்குறட்பாவில் பயின்று வந்துள்ள அணி உவமை அணி.
அணி இலக்கணம்:
செய்யுளில் உவமை ஒரு வாக்கியமாகவும், உவமேயம் மற்றொரு வாக்கியமாகவும், உவம உருபு வெளிப்படையாக வருவது உவமை அணி ஆகும்.
உவமை : மலைமேல் பாதுகாப்பாக நின்று யானைப் போரைக் காணுதல்.
உவமேயம் : தன் கைப்பொருளைக் கொண்டு செய்யும் செயல்.
உவம உருபு : அற்று (வெளிப்படை)
பொருத்தம்: தன் கைப்பொருளைக் கொண்டு ஒருவர் ஒரு செயலைச் செய்வது மலைமேல் பாதுகாப்பாக நின்று கொண்டு யானைப் போரைக் காண்பது போன்றது ஆகும்.
![]()
Question 5.
இன்மையின் இன்னாத தியாதெனின் இன்மையின்
இன்மையே இன்னா தது – இக்குறட்பாவில் பயின்றுவரும் அணியைக் கூறி விளக்குக.
Answer:
இக்குறட்பாவில் பயின்று வந்துள்ள அணி சொற்பொருள் பின்வருநிலையணி.
அணி இலக்கணம்:
ஒரு செய்யுளில் முன்னர் வந்த சொல் மீண்டும் மீண்டும் வந்து ஒரே பொருளைத் தருவது சொற்பொருள் பின்வருநிலையணி ஆகும்.
பொருள் விளக்கம்: இக்குறட்பாவில் இன்மை என்ற சொல் மீண்டும் மீண்டும் வந்து வறுமை என்னும் ஒரே பொருளைத் தருகிறது. எனவே, இக்குறட்பா சொற்பொருள் பின்வருநிலையணி ஆயிற்று.
Question 6.
தேவர் அனையர் கயவர் அவரும்தாம்
மேவன செய்தொழுக லான் – இக்குறட்பாவில் பயின்றுவந்துள்ள அணியைச் சுட்டி விளக்குக.
Answer:
இக்குறட்பாவில் பயின்று வந்துள்ள அணி வஞ்சப் புகழ்ச்சி அணி.
அணி இலக்கணம்:
பழிப்பது போலப் புகழ்வதும் புகழ்வது போலப் பழிப்பதும் வஞ்சப்புகழ்ச்சி அணி ஆகும். இப்பாடல் புகழ்வது போலக் கூறி பழிக்கப்பட்டுள்ளது.
விளக்கம் :
புகழ்தல் – கயவர் தேவர்களுக்கு ஒப்பாவர்
பழித்தல் – கயவர் தம் மனம் போன போக்கில் தீய செயல்களைச் செய்வார்கள்.
தேவரும் கயவரும் ஒரு தன்மையர். தேவரைப் போல கயவரும் தாம் விரும்பியதைச் செய்வர் என்று புகழ்வது போல பழித்தலால், இஃது வஞ்சப்புகழ்ச்சி அணி ஆயிற்று.
Question 7.
மக்களே போல்வர் கயவர்; அவரன்ன
ஒப்பாரி யாம்கண்ட தில் – இப்பாடலில் பயின்று வந்துள்ள அணியைக் கூறி விளக்குக.
Answer:
இக்குறட்பாவில் பயின்று வந்துள்ள அணி உவமை அணி ஆகும்.
அணி இலக்கணம்:
செய்யுளில் உவமை ஒரு வாக்கியமாகவும், உவமேயம் மற்றொரு வாக்கியமாவும், உவம உருபு வெளிப்படையாக வருவது உவமை அணி ஆகும்.
உவமை : கயவர் மக்களைப் போல் இருப்பர்.
உவமேயம் : தோற்ற ஒப்புமையைத் தவிர வேறெந்த ஒப்புமையும் இல்லை.
உவம உருபு : அன்ன (வெளிப்படை)
விளக்கம் : கயவர் மக்களைப் போலவே இருப்பர்; கயவருக்கும் மக்களுக்கும் உள்ள தோற்ற ஒப்புமையை வேறெதிலும் காண்பதில்லை.
![]()
நெடுவினா
Question 1.
பொருள் செயல்வகை என்னும் அதிகாரத்தில் வள்ளுவர் கூறும் வளமார்ந்த கருத்துக்களைக் கூறுக.
Answer:
சிறந்த பொருள் :
ஒரு பொருட்டாக மதிக்கத் தகாதவரையும் மதிப்புடையவராகச் செய்வது செல்வம். செல்வத்தை அல்லாமல் சிறந்த பொருள் உலகில் வேறு எதுவும் இல்லை .
அறம், இன்பம்:
முறையறிந்து தீமையற்ற வழியில் ஒருவர் சேர்த்த செல்வம் அவருக்கு அறத்தையும் தரும், இன்பத்தையும் தரும்
நீக்கவேண்டிய பொருள்:
மற்றவர்களிடம் இரக்கமும் அன்பும் இல்லாமல் ஈட்டும் பொருளை ஏற்றுக்கொள்ளாமல் நீக்கிவிட வேண்டும்.
தன் கைப்பொருள்:
தன் கைப்பொருளைக் கொண்டு ஒருவன் செய்யும் செயலானது, குன்றின் மேலே பாதுகாப்பாக நின்று கொண்டு யானைப் போரைக் காண்பதற்குச் சமமாகும்.
![]()
கூர்மையான ஆயுதம்:
பொருளை ஈட்ட வேண்டும். பகைவரை வெல்லும் கூர்மையான ஆயுதம் பொருளைத் தவிர வேறு ஒன்றும் இல்லை .