Students get through the TN Board 12th Chemistry Important Questions Chapter 3 p-Block Elements – II which is useful for their exam preparation.
TN State Board 12th Chemistry Important Questions Chapter 3 p-Block Elements – II
Answer the following questions.
Question 1.
Briefly account for the trend in atomic radius of elements in the nitrogen family.
Answer:
Atomic radii increase on going down the group. The increase is due to addition of new energy level in each succeeding element. There is a considerable increase in covalent radius from N to P but from As to Bi only a small increase is observed.
The increase in nitrogen to phosphorus is attributed to strong shielding effect of ‘s’ and ‘p’ electrons present in inner shells. Small increase in covalent radii from As to Bi is due to poor shielding effect of ‘d’ and ‘f’ electrons present in inner shells on valency electrons. The increased nuclear charge reduces the effect of the addition of new shell to same extent.
Question 2.
Briefly explain the trend in melting and boiling point in the nitrogen family elements.
Answer:
The melting point first increase from nitrogen to arsenic and then decreases from antimony and bismuth. The boiling point gradually increases from nitrogen to antimony. Lower values of melting points for antimony and bismuth may be due to availability of only three electrons instead of five for metallic bonding due to inert pair effect.
![]()
Question 3.
What happens when (i) Sodium azide is heated? (ii) Ammonia is treated with bromine. Give equations.
Answer:
In both cases, nitrogen is formed.
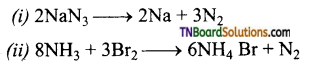
Question 4.
Explain why nitrogen is inert at room temperature.
Answer:
This is due to high bond enthalpy of N ≡ N. The triple bond is short and hence large amount of heat is necessary to break the bond. Hence N2 is unreactive at room temperature.
Question 5.
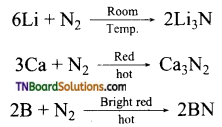
What are nitrides? Give the preparation of (i) Lithium nitride, (ii) Calcium nitride, (iii) Boron nitride by means of chemical equation.
Answer:
Nitrides are binary compounds of nitrogen which contains N-3 ion. They are formed by direct combination of metals with nitrogen.
Question 6.
Mention the conditions under which maximum amount of ammonia is formed from nitrogen to hydrogen.
Answer:
The formation of NH3 is represented by
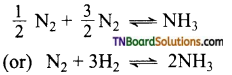
is an exothermic reaction. The reaction is favoured at high temperatures and at an optimum temperature in the presence of iron as catalyst.
Question 7.
Give equation for (i) hydrolysis of urea,
(ii) heating ammonium chloride with CaO,
(iii) Heating magnesium nitride with water.
Answer:
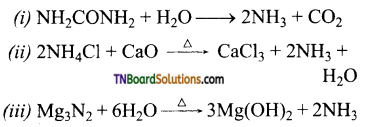
![]()
Question 8.
How is ammonia manufactured?
Answer:
Ammonia is manufactured by passing nitrogen and hydrogen over iron catalyst (a small amount of K2O and Al2O3 is also used to increase the rate of attainment of equilibrium) at 750 K at 200 atm pressure. In the actual process the hydrogen required is obtained from water gas and nitrogen from fractional distillation of liquid air.
Question 9.
Compare the properties of liquid ammonia and water.
Answer:
(i) Both liquid ammonia and water are highly associated through strong hydrogen bonding.
(ii) Both ammonia and water are good ionising solvents.
(iii) The ionisation of ammonia and water is given as
![]()
![]()
Both OH– and NH2– are strong bases,
eg: NaOH and NaNH2.
Question 10.
Give equations for the following reactions.
(i) Ammonia is heated over 500°C
(ii) Ammonia is burnt in oxygen.
(iii) Ammonia is burnt in oxygen in the presence of a metal catalyst (pt)
(iv) Ammonia is treated with excess of chlorine.
(v) Excess of ammonia is treated with chlorine.
Answer:
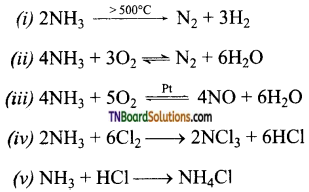
Question 11.
Give an example for a reducing property of ammonia. [OR] Ammonia is a reducing agent. Give an example to prove this statement.
Answer:
It reduces metal oxides to metals when passed over heated metallic oxide.
![]()
In this reaction, ammonia is oxidised to nitrogen.
Question 12.
What are amides and nitrides? Give an example for each. How are they formed?
Answer:
Amides are salts formed by ammonia and a strong electropositive metal like Na, eg: NaNH2 (sodamide). It is formed by the action of ammonia on sodium.
2Na + 2NH3 → 2NaNH2 + H2
Nitrides are salts which contain N-3 ion.
eg: Mg3N2 (magnesium nitride). It is formed by the action of magnesium with ammonia.
3Mg + 2NH3 → Mg3 N2 + 3H2
![]()
Question 13.
What happens when an aqueous solution of ammonia is treated with (i) aqueous solution of ferric chloride, (ii) an aqueous solution of cupric chloride, (iii) an aqueous solution of aluminium chloride.
Answer:
An aqueous solution of ammonia or ammonium hydroxide precipitates metal hydroxides from metallic salts solution.
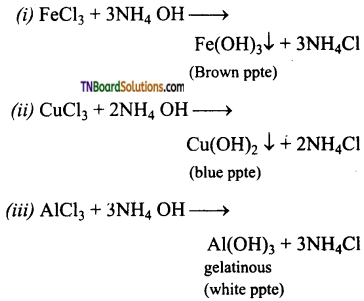
This property is made use of in detecting group III metals in qualitative analysis.
Question 14.
Explain with examples, that ammonia acts as a Lewis base.
Answer:
Due to the presence of lone pair of electrons on the nitrogen atom, ammonia acts as a Lewis base. It can donate its electron pair to form coordinate covalent bond with electron deficient molecules eg: BF3 or transition metal cation, having vacant ‘d’ orbitals) to form complexes. For example,
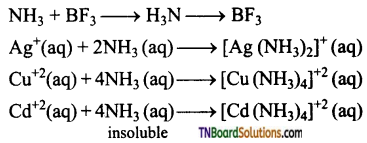
Question 15.
Explain why (i) insoluble silver chloride dissolves in aqueous ammonia?
Answer:
This is due to the formation of a complex Ag (NH3)2 Cl (diammine silver (I) chloride)
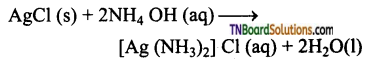
Question 16.
When aqueous ammonia is treated with a copper sulphate solution, a blue precipitate is formed It dissolves an adding excess ammonia. Explain this observation.
Answer:
The precipitate formed is cupric hydroxide, which dissolves in excess ammonia to form a blue solution due to the formation of soluble [Cu (NH3)4] SO4 complex, i.e., tetrammine copper(II) sulphate.
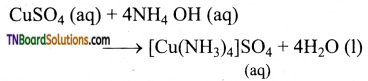
Question 17.
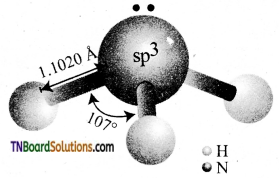
Explain the structure of ammonia.
Answer:
The nitrogen in ammonia is sp3 hybridised. Each of the sp3 hybrid orbital containing one unpaired electron forms a covalent bond with the 1s orbital of hydrogen. The fourth sp3 hybrid orbital contain a lone pair of electrons. Because of the lone pair – bond pair repulsion, the tetrahedral bond angle is reduced to 107°. Hence, it assumes a pyramidal shape.
![]()
Question 18.
How is nitric acid prepared?
Answer:
Nitric acid is prepared by heating equal amounts of potassium or sodium nitrate with concentrated sulphuric acid.
KNO3 + H2SO4 → KHSO4 + HNO3
The temperature is kept as low as possible to avoid decomposition of nitric acid.
Question 19.
Give reason for the following:
Answer:
The nitric acid prepared by heating potassium nitrate and conc. H2SO4 is brown coloured.
The acid formed by heating KNO3 and H2SO4 dioxide formed by the decomposition of nitric acid.
4HNO3 → 4NO2 + 2H2O + O2
Question 20.
Explain the manufacture of Oswalld’s process of nitric acid.
Answer:
The method is based as upon the catalystic oxidation of ammonia by atmospheric oxygen.
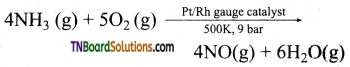
Nitric oxide thus formed readily combines with oxygen to form nitrogen dioxide (NO2).
2NO(g) + O2(g) → 2NO2(g)
Nitrogen dioxide, thus formed dissolves in water to form nitric acid.
3NO2 (g) + H2O (l) → 2HNO3 (aq) + NO (s)
The nitric oxide formed is recycled and the aqueous nitric acid is concentrated by distillation to give 68% nitric acid by mass. Further concentration to 98% can be achieved by dehydration with conc. H2SO4.
Question 21.
Explain with examples to show that nitric acid acts as an (i) acid, (ii) oxidising agent and (iii) as nitrating agent.
Answer:
(i) It reacts with bases and basic oxides to form salts and water.

(ii) It oxidises non metals to their highest oxy acids.
(iii) As a nitrating agent, it forms nitronium ion (NO2+) in aromatic electrophillic substitution reactions.

Question 22.
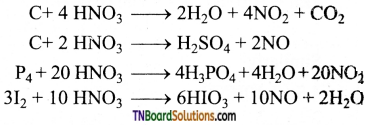
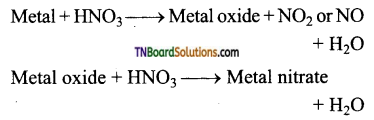
Briefly explain the action of nitric acid on metals.
Answer:
Nitric acid, both dilute and concentrated are powerful oxidising agents and metals are good reducing agents. The products of oxidation depending on the concentration of the acid and the nature of the metal.
(i) Gold, platinum, rhodium, iridium and tantalum do not react with nitric oxide.
(ii) Metals which are more electropositive than hydrogen (Na, K, Ca, Mg, Al, Mn, Zn, Cr, Cd, Fe, CO, Ni, Sn, Pb, etc.,) reduces HNO3 to a variety of products such as NO2, NO, NH3, NH4 NO3 and N2O, depending upon the conditions.
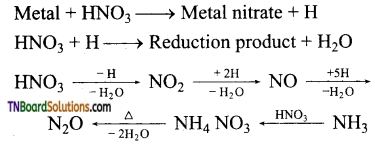
(iii) Magnesium and manganese are the only metals that produce H2 with very dilute (1-2%) HNO3.
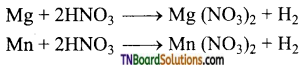
(iv) More active metals like magnesium, zinc, tin and iron react with cold, dilute nitric acid to form ammonium nitrate.

Lead, under similar condition, give lead nitrate and nitric oxide.

(v) With hot, dilute nitric acid, ammonium nitrate thus formed undergoes decomposition to form nitrous oxide (N2O)
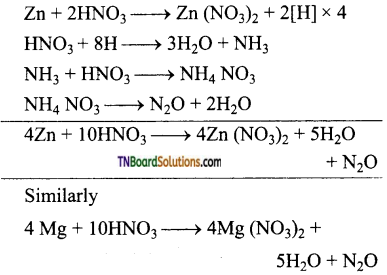
(vi) Metals like Zn, Mg, Bi, Pb, etc., form metallic nitrate and nitrogen dioxide when treated with conc. HNO3.
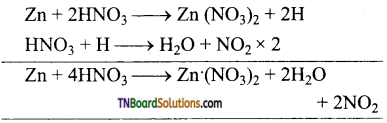
(vii) Similarly,

Exception: Tin forms metastannic acid H2SnO3 and NO2
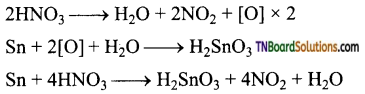
(viii) Metals which are less electropositive than hydrogen, reduce nitric acid to NO2 or NO.
eg:
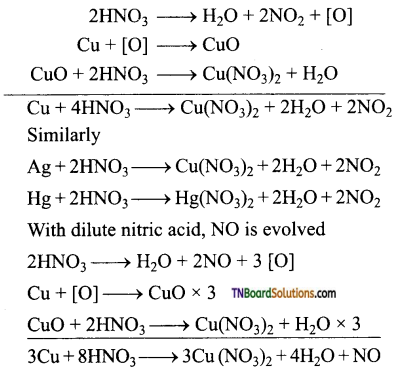
(ix) Metals like iron, chromium, nickel and aluminium because passive as treatment with cone. HNO3.
(x) Noble metals like gold, platinum do not react with conc. HNO3.
Question 23.
Give uses of nitric acid.
Answer:
- Nitric acid is used as a oxidising agent and in the preparation of aquaregia.
- Salts of nitric acid are used in photography (AgNO3) and gunpowder for firearms. (NaNO3).
![]()
Question 24.
Complete and balance the following equations.
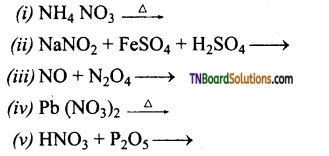
Answer:
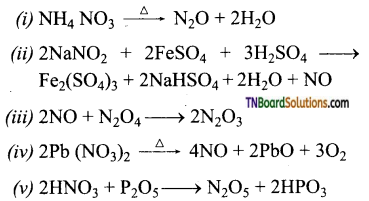
Question 25.
Give the oxidation states of nitrogen in the following oxides.
(i) N2O, (ii) NO, (iii) N2O3, (iv) NO2, (v) N2O4, (vi) N2O5.
Answer:
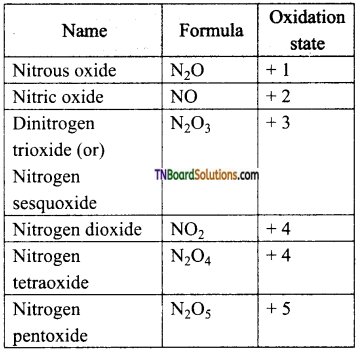
Question 26.
Write the structure of the following oxides of nitrogen, (i) N2O, (ii) NO, (iii) N2O3, (iv) NO2, (v) N2O4, (vi) N2O5.
Answer:
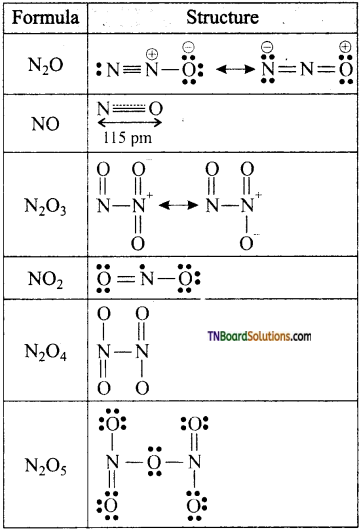
Question 27.
Give equation for the preparation of the following acids.
(i) Hyponitrous acid,
(ii) Nitrous acid,
(iii) Pemitrous acid,
(iv) Nitric acid,
(v) Pemitric acid.
Answer:
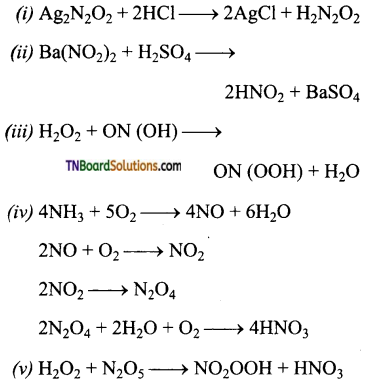
![]()
Question 28.
Give reason for the following:
(i) Freshly prepared phosphorus becomes yellow on standing.
(ii) Yellow phosphorus glows in dark.
(iii) Nitrogen is a gas while phosphorus is a solid.
Answer:
(i) This is due to the formation of a red phosphorus upon standing.
(ii) This is due to oxidation which is known as phosphorus.
(iii) The nitrogen has N ≡ N structure while in phosphorus four atoms in phosphorus have a polymeric structure with chains of P4 linked tetrahedrally. P ≡ P is less stable than P — P bonds. Hence nitrogen is a gas while phosphorus is a solid.
Question 29.
Write the structure of the following:
(i) Hyponitrous acid, (ii) Hydronitrous acid, (iii) Nitrous acid, (iv) Pemitrous acid, (v) Nitric acid, (vi) Pemitric acid.
Answer:
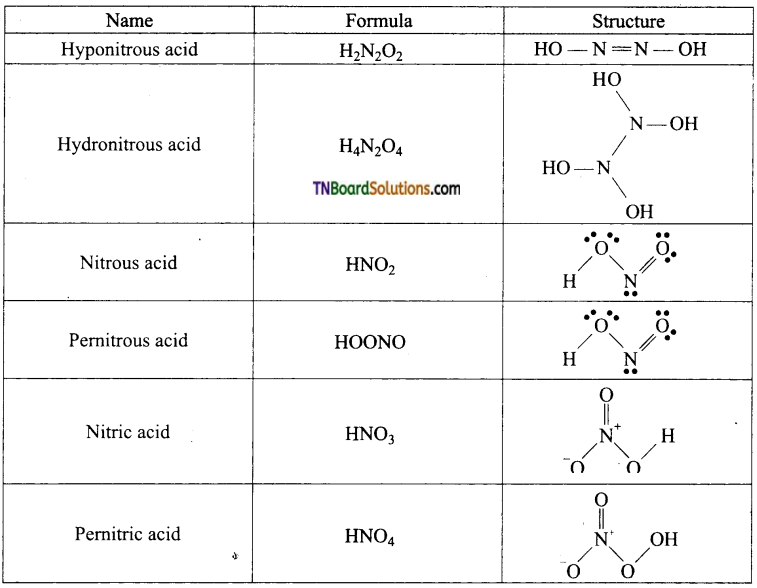
Question 30.
Give the formula and the oxidation state of nitrogen in the following acids.
(i) Hyponitrous acid, (ii) Nitrous acid, (iii) Pemitrous acid, (iv) Nitric acid, (v) Pemitric acid.
Answer:
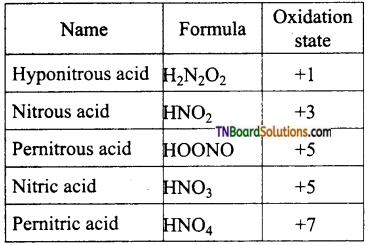
Question 31.
Explain the structure of white phosphorus.
Answer:
It exists as P4 units. The four sp3 hybridised phosphorus atoms lie at the corners of a regular tetrahedral with ∠PPP=60° as shown in figure.
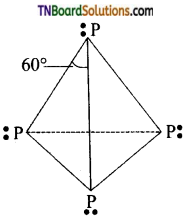
Each phosphorus atom is linked to other three phosphorus atoms by covalent bonds so that each P-atom completes its octet.
![]()
Question 32.
Explain why white phosphorus is kept under water.
Answer:
The P4 units of white phosphorus are held together by weak vanderwaals forces of attraction. As a result, its ignition temperature (303K) is very low and easily catches fire. Hence, it is kept under water.
Question 33.
The red phosphorus is less reactive than white phosphorus. Explain with examples.
Answer:
(i) Yellow phosphorus readily catches fire in air forming phosphorus pentoxide whereas red phosphorus forms phosphorus trioxide or phosphorus pentoxide only on heating.
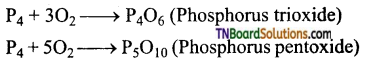
(ii) With chlorine, phosphorus forms phosphorus tri and penta chlorides. White phosphorus reacts violently at room temperature while red phosphorus react on heating.
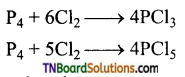
(iii) Yellow phosphorus reacts with alkali an boiling in an inert atmosphere to form phosphine. Red phosphorus has no action an alkali.
Question 34.
Distinguish between white and red phosphorus interms of their properties.
Answer:
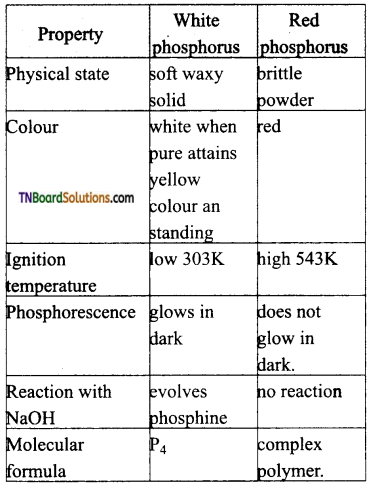
Question 35.
Compare the chemical properties of white and red phosphorus.
Answer:
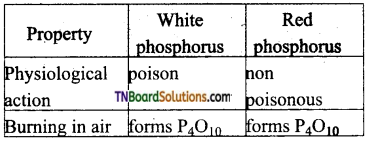
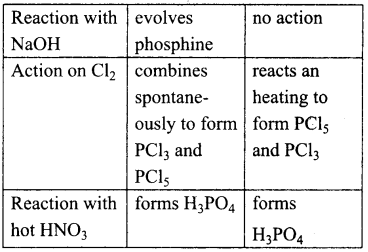
Question 36.
How phosphine is formed from (i) White phosphorus, (ii) Calcium phosphide, (iii) Phosphorus acid, (iv) Phosphonium iodide. Give equations.
Answer:
(i) By heating white phosphorus and sodium hydroxide in an inert atmosphere of carbon dioxide or hydrogen.
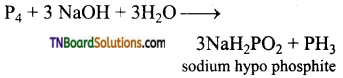
(ii) By the hydrolysis of calcium phosphide with water or dilute mineral acids.
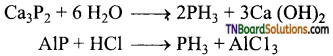
(iii) By heating phosphorus acid (H3PO3)
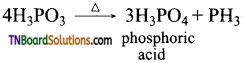
(iv) By heating phosphorus iodide with sodium hydroxide.
![]()
![]()
Question 37.
Give equations for the following:
(i) Phosphine is heated at 317 K in the absence of air.
(ii) Phosphine is heated with oxygen.
(iii) Phosphine is treated with chlorine.
Answer:
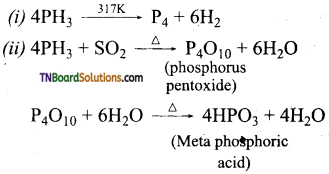
(iii) PH3 + 4Cl3 → PCl5 + 3HCl
Question 38.
Give examples for (i) basic nature and (ii) reducing property of phosphine.
Answer:
(i) Basic nature of PH3: Because it contains a lone pair of electrons as phosphorus, it accepts a proton and form phosphorium ion.
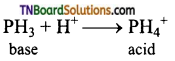
It is a weaker base than NH3 because of larger size and lesser electronegativity of phosphorus than nitrogen.
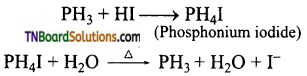
(ii) Reducing property of PH3: Phosphine reduces aqueous solutions of copper and mercury salts to their corresponding phosphides.
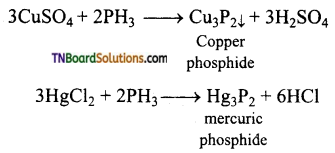
Question 39.
Briefly explain the structure of phosphine.
Answer:
The phosphorus in phosphine is sp3 hybridised. The three sp3 hybrid orbitals each containing an electron overlaps with 1s orbital of hydrogen, containing are electron form three P — H covalent bonds. The fourth sp3 hybrid orbital contains a lone pair of electron. The lone pair-bond pair interaction, due to the larger size and lesser electro negativity of phosphorus, compared to nitrogen, reduces the tetrahedral bond angle to 94°. Hence phosphine has a pyramidal shape, like ammonia.
Question 40.
What are Holmes signals? Mention its use.
Answer:
Phosphine (PH3) is used in Holme’s signals in deep seas and oceans for signalling danger points to steamers. Containers containing a mixture of calcium phosphide and calcium carbide are pierced and thrown into the sea.
In contact with water, a mixture of acetylene and phosphine is produced. Phosphine contains traces of highly inflammable P2H4 which catches five spontaneously. This ignites acetylene with a luminous flame and thus serves as a signal to the approaching ship.
Question 41.
How is phosphorus trichloride prepared? [OR] What happens when white phosphorus is treated with a stream of chlorine gas.
Answer:
P4 + 3Cl2 → P4Cl6
![]()
Question 42.
What happens when (i) White phosphorus is treated with thionyl chloride.
(i) White phosphorus is treated with thionyl chloride.
(ii) Phosphorus trichloride is hydrolysed by cold water.
Answer:
(i) Phosphorus trichloride is formed.
4P4 + 8SOCl2 → 4PCl3 + 4SO2 + 2S2Cl2
(ii) Hydrolysis of phosphorus trichloride gives phosphorus acid. (H3PO3)
PCl3 + 3H2O → H3PO3 + 3HCl
Question 43.
How does phosphorus trichloride react with
(i) ethanol and (ii) propionic acid? Give equations.
Answer:
The ‘OH’ group in alcohols and acids are replaced by ‘Cl’.
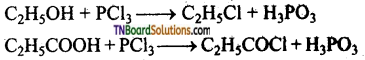
Question 44.
Explain the structure of phosphorus trichloride.
Answer:
Its structure is similar to ammonia. P atoms undergoes sp3 hybridisation. In the tetrahedral configuration one of the positions is occupied by the lone pair. Thus, it is pyramidal in shape. The Cl — P — Cl bond angle is 100.4° and P — Cl bond length is 204 pm.
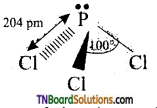
Question 45.
Give equations for (i) the formation of PCl5 from PCl3 (ii) H3PO4 from PCl5.
Answer:
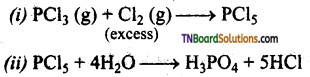
Question 46.
How does phosphorus pentachloride react with metals? Give examples.
Answer:
Phosphorus penta chloride (PCl5) reacts with metals to give metal chlorides.

Question 47.
Briefly outline the structure of PCl5.
Answer:
It has a trigonal bipyramidal shape in which P undergoes sp3d hybridisation.
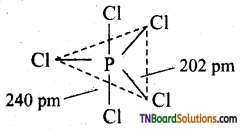
Question 48.
Mention the uses of phosphorus pentachloride.
Answer:
Phosphorus pentachloride is a chlorinating agent and is useful for replacing hydroxyl groups by chlorine atom.
![]()
Question 49.
Hypophosphorus acid is monobasic and a good reducing agent. Explain.
Answer:
The structure of hypophosphorus acid is
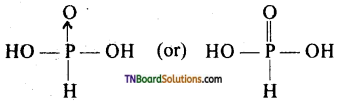
It has One P — OH bond, two P — H and P → O or P = O bonds. Of the three hydrogen atoms, only one hydrogen atom attached to the oxygen atom (OH group) is replaceable. Hence it is a monobasic acid. Because of the presence of P — H bond, it acts as a reducing agent.
Question 50.
Briefly explain the structure of phosphorus trioxide and phosphorus pentoxide.
Answer:
(i) Structure of phosphorus trioxide: The phosphorus atom lie at tetrahedral positions with respect to each other and six oxygen atoms are inserted between them. Each phosphorus atom is covalently bonded to three oxygen atoms and each oxygen is bonded to two phosphorus atom. The P — O bond length is 165.6 pm.
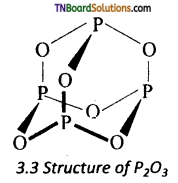
(ii) Structure of phosphorus pentoxide: In P4O10, each P atom form three bonds with oxygen atom an also an additional coordinate bond with an oxygen atom. Terminal coordinate P — O bond length is 143 pm.
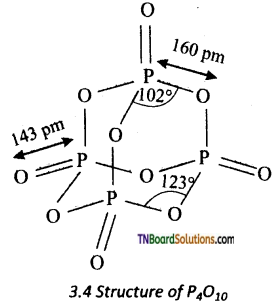
Question 51.
What is the basicity of orthophosphorus acid? Explain with its structure. Will it act as a reducing agent or not?
Answer:
The structure of orthophosphorus acid is
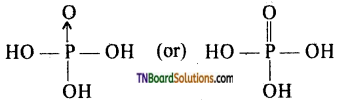
It has two P — P — OH bonds, one P → O or P = O bond, and one P — H bond. There are two ‘OH’ groups attached to the phosphorus atom, and the hydrogen atoms are replaceable.
Hence it is a dibasic acid. The acid and their salts are good reducing agent because of P —H bond.
Question 52.
Write the formula and structure of hypo phosphoric acid. What information do you get from its structure regarding its basicity and its reducing action?
Answer:
The formula is H4P2O6 and its structure is
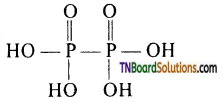
It contains ‘4’ OH group and 4 replaceable hydrogen atoms. Hence it is a tetrabasic acid or its basicity is four. It does not contain a P — H bond. Hence, it does not act as a reducing agent.
Question 53.
Orthophosphoric acid is tribasic and is not a reducing agent. Explain.
Answer:
Orthophosphoric acid (H3PO4) is tribasic, because it contains three OH groups attached to the phosphorus atom where all the hydrogens are replaceable.
Hence, it is a tribasic acid.
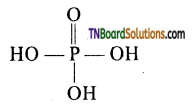
It forms three series salt, by successive replacement of H atom of the ‘OH’ group,
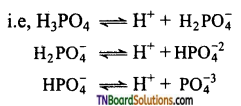
These salts are known as dihydrogen phosphate, hydrogen phosphate and phosphate respectively. It is not a reducing agent because it does not contain a P — H bond.
![]()
Question 54.
Write the structure of pyrophosphoric acid and explain its basicity on the basis of the structure.
Answer:
Phosphoric acid (H4P2O7) has the structure
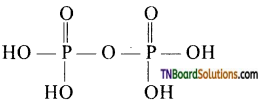
It is a tetra basic acid.
Question 55.
Write the formula and the oxidation state of phosphorus in the following oxyacids.
(i) Hypophosphorus acid
(ii) Orthophosphorus acid
(iii) Hypophosphoric acid
(iv) Orthophosphoric acid
(v) Pyrophosphoric acid
Answer:

Question 56.
Complete and balance the following equations:
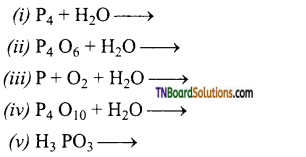
Answer:
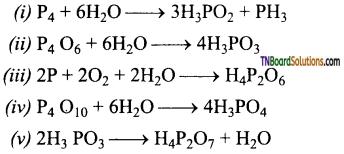
Question 57.
Briefly explain the trend in physical properties of group 16 elements.
Answer:
Group 16 elements consist of oxygen (O), sulphur (S), selenium (Se), tellurium (Te) and polonium (PO). They are also called oxygen family or chalcogens, because many metals occur as oxides and sulphides.
- The electronic configuration of these elements are:

- The atomic and ionic radii of group 16 elements are smaller than those of the corresponding elements of group 15. Further, the atomic radii increases down the group. The increase in atomic radii of group 16 elements in primarily due to increase in the number of electron shells.
- The melting, boiling points and densities increase regularly as we go down the group upto tellunium. However, the melting and boiling points of polonium are lower than those of tellurium. This is because, the atomic size increases down the group. As a result, vanderwaals forces of attraction among their atoms also increase and hence melting and boiling points regularly increase from oxygen to tellurium. However due to presence of inner ‘d’ and ‘f’ electrons, the inert pair effect is maximum in polonium. Consequently, the ‘s’ valence electrons in polonium are less available as compared to those in tellurium for bonding. As a result, vanderwaals forces of attraction will be weaker in Po than in Te and therefore melting point and boiling point of Po will be lower than that of Te.
- Oxygen exists as a diatomic gas while other elements exist as octa atomic solids.
- All the elements of this group show allotropy. Oxygen exists in two non metallic forms viz dioxygen (O2), tri oxygen O3. Sulphur exists in crystalline as well as amorphous allotropic forms. The crystalline form includes rhombic sulphur (α sulphur) and mono clinic sulphur (β sulphur). Amorphous allotropic forms include plastic sulphur (γ sulphur), milk of sulphur, and colloidal sulphur.
![]()
Question 58.
Give two methods of preparation of oxygen in the laboratory.
Answer:
In the laboratory, oxygen is prepared by one of the following methods.
The decomposition of hydrogen peroxide in the presence of a catalyst (MnO2) or by oxidation with potassium permanganate.
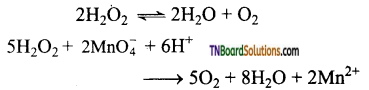
The thermal decomposition of certain metallic oxides or oxoanions gives oxygen.
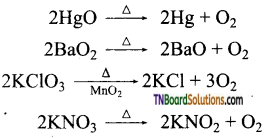
Question 59.
Give reasons for
(i) Oxygen exists as a diatomic gas.
(ii) Oxygen is paramagnetic.
(iii) Oxygen forms strong hydrogen bonds.
Answer:
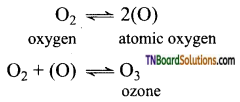
(i) Due to small size and high electronegativity, oxygen forms pπ — pπ, double bond with another oxygen atom to form O = O molecule. The intermolecular forces of attraction between oxygen molecules are weak and hence oxygen exists as a diatomic gas.
(ii) The oxygen is paramagnetic because it contains two unpaired electrons in the antibonding molecular orbitals.
(iii) The high electronegativity of the oxygen atom is responsible to form hydrogen bonding eg: H2O.
Question 60.
How is ozone prepared in the laboratory?
Answer:
Ozone is prepared in the laboratory by passing silent electrical discharge through oxygen. At a potential of 20,000 V about 10% of oxygen is converted into ozone it gives a mixture known as ozonised oxygen. Pure ozone is obtained as a pale blue gas by the fractional distillation of liquefied ozonised oxygen.
Question 61.
Write a note on the structure of ozone molecule.
Answer:
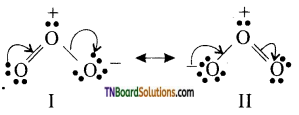
The central oxygen atom in ozone is sp2 hybridised containing a lone pair of electrons. As a result, ozone has an angular structure.
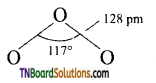
It is actually a resonance hybrid of the following two resonating structural (I and II) as shown is figure.
Question 62.
How does oxygen react with metals and non metals? Give examples.
Answer:
Oxygen reacts with metals and non metals and form oxides.
(i) Oxygen combines with many metals and non metals to form oxides.
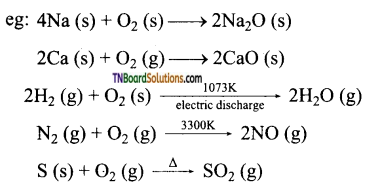
(ii) Some less reactive metals react when powdered finely and made to react exothermically with oxygen at room temperature.
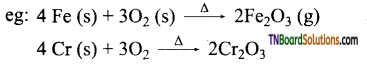
Question 63.
Explain why ozone is a more powerful
oxidising agent than oxygen.
Answer:
This is due to the reason that ozone has higher energy content than oxygen and undergo decomposition.
2O3 (g) → O2 (g) + [O]
The atomic oxygen, thus liberated brings about oxidation.
![]()
Question 64.
Give an example for the oxidising action of ozone.
Answer:
Ozone oxidises potassium iodide to iodine.
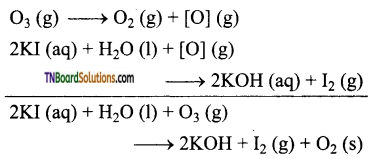
Question 65.
Explain how ozone can be estimated quantitatively.
Answer:
Ozone gas is passed through an aqueous solution of potassium iodide. The liberated iodine is titrated with a standard solution of theo sulphate. From the volume of the standard solution of sodium theo sulphate, the amount of ozone can be calculated.
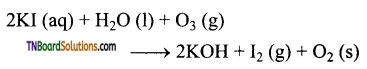
Question 66.
Mention the uses of oxygen.
Answer:
- Oxygen is one of the essential components for the survival of living organisms.
- It is used in welding (oxy acetylene welding).
- Liquid oxygen is used as fuel in rockets etc.
Question 67.
Name the stable allotrope of sulphur at ordinary temperature and pressure. What happens when it is heated to avoid 96°C?
Answer:
Rhombic sulphur is the most stable allotrope of sulphur at ordinary temperature and pressure. When heated slowly at 96°C. It is converted to monoclinic sulphur.
Question 68.
Give the characteristics of rhombic and monoclinic sulphur.
Answer:
Rhombic sulphur consist of yellow coloured crystals and made up of S8 molecules. Monoclinic sulphur also contains S8 molecules in addition to S6 molecules (small amount). It exists as a long needle like prism. It is stable between 96 – 119°C.
Question 69.
How is sulphur dioxide prepared from
(i) sulphur, (ii) galena, (iii) iron pyrites.
Answer:
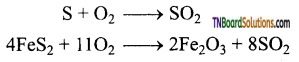
(i) Burning sulphur in air.
S + O2 → SO2

Question 70.
Give equation for the preparation of sulphur dioxide in the laboratory.
Answer:
![]()
![]()
Question 71.
With an example, prove sulphur dioxide in an acidic oxide.
Answer:
It dissolves in water producing sulphurous acid. (H2SO3)

It reacts with sodium hydroxide and sodium carbonate to form sodium hydrogen sulphate and sodium sulphate respectively.
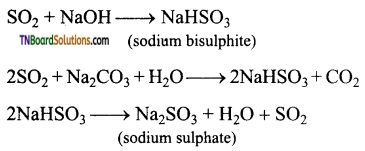
Question 72.
Give two examples for the oxidising property of sulphur dioxide.
Answer:
It oxidises hydrogen sulphide to sulphur and magnesium to magnesium oxide.
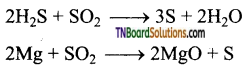
Question 73.
Give two examples to show that sulphur dioxide acts as a reducing agent.
Answer:
(i) It reduces chlorine water to hydrochloric acid.
![]()
(ii) It reduces potassium permanganate and potassium dichromate to Mn+2 and Cr+3 respectively.
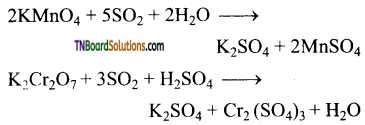
Question 74.
Give equation for the reaction in which sulphur dioxide is used for the manufacture of sulphuric acid by contact process.
Answer:
![]()
Question 75.
Explain the use of sulphur dioxide as a bleaching agent.
Answer:
In presence of water, sulphur dioxide bleaches coloured wool, silk, sponges and straw into colourless due to its reducing property.
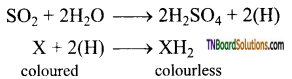
However, the bleached product (colourless) is allowed to stand in air, it is reoxidised by atmospheric oxygen to its original colour. Hence bleaching action of sulphur dioxide is temporary.
![]()
Question 76.
Draw the structure of sulphur dioxide.
Answer:
In sulphur dioxide, sulphur atom undergoes sp2 hybridisation. A double bond arises between S and O is due to pπ- dπ overlapping.
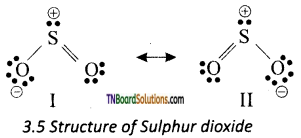
It is a resonance hybrid of the canonical form I and II.
Question 77.
Mention the uses of sulphur dioxide.
Answer:
- Sulphur dioxide is used in bleaching hair, silk, wool etc…
- It can be used for disinfecting crops and plants in agriculture.
Question 78.
Discuss the various steps involved in the manufacture of sulphuric acid by contact process?
Answer:
The contact process involves the following steps.
- Initially sulphur dioxide is produced by burning sulphur or iron pyrites in oxygen/air.
- Sulphur dioxide formed is oxidised to sulphur trioxide by air in the presence of a catalyst such as V2O5 or platinised asbestos.
- The sulphur trioxide is absorbed in concentrated sulphuric acid and produces oleum (H2S2O7). The oleum is converted into sulphuric acid by diluting it with water.
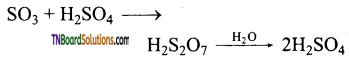
To maximise the yield the plant is operated at 2 bar pressure and 720 K. The sulphuric acid obtained in this process is over 96 % pure.
Question 79.
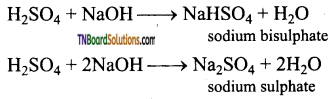
How do you prove that sulphuric acid is dibasic acid?
Answer:
The structure of sulphuric acid is
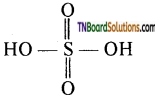
It has two replaceable hydrogen atoms. Hence, it forms two types of salts viz bi sulphates and sulphates. Hence H2SO4 is a dibasic acid.
![]()
Question 80.
Give two examples to show that cone, sulphuric acid is an oxidising agent.
Answer:
(i) It oxidises carbon to carbon dioxide and gets reduced to sulphur dioxide
![]()
(ii) It oxidises hydrogen sulphide to sulphur. It gets reduced to sulphur dioxide.
![]()
Question 81.
Complete and balance the following equations.
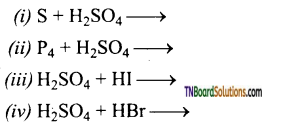
Answer:

Question 82.
Give a brief account of the action of sulphuric acid on metals.
Answer:
Sulphuric acid reacts with metals and gives different products depending on the reactants and reacting condition. Dilute sulphuric acid reacts with metals like tin, aluminium, zinc to give corresponding sulphates.

Hot concentrated sulphuric acid reacts with copper and lead to give the respective sulphates as shown below.

Sulphuric acid doesn’t react with noble metals like gold, silver and platinum.
Question 83.
What happens when
(i) Potassium chloride is heated with concentrated sulphuric acid.
(ii) Potassium nitrate is heated with concentrated sulphuric acid.
(iii) Sodium carbonate is heated with dilute sulphuric acid.
(iv) Sodium bromide is heated with cone, sulphuric acid?
Answer:
(i) Hydrogen chloride is produced.
KCl + H2SO4 → KHSO4 + HCl
(ii) Sulphuric acid, being a strong acid displaces relatively weaker nitric acid from its salt.
KNO3 + H2SO4 → KHSO4 + HNO3
(iii) It decomposes sodium carbonate to carbon dioxide and water.
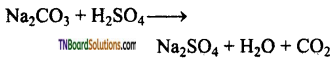
(iv) It oxidises Br– to Br2.
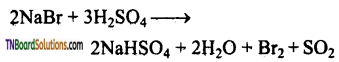
![]()
Question 84.
How will you detect sulphate radical in qualitative analysis.
Answer:
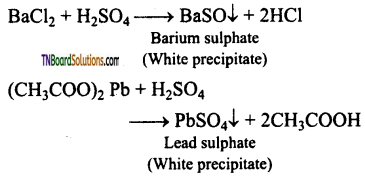
Dilute solution of sulphuric acid/aqueous solution of sulphates gives white precipitate (barium sulphate) with barium chloride solution. It can also be detected using lead acetate solution. Here a white precipitate of lead sulphate is obtained.
Question 85.
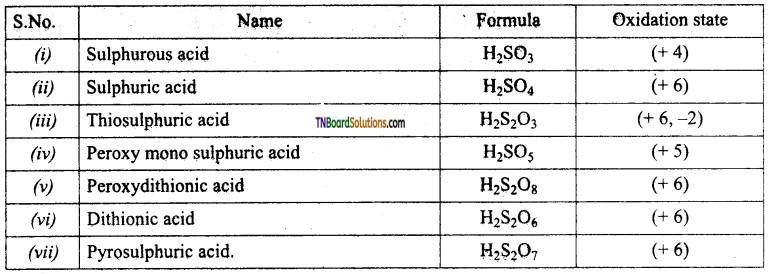
Write the formula and the oxidation state of sulphur in the following oxoacids.
(i) Sulphurous acid,
(ii) Sulphuric acid,
(iii) Thio sulphuric acid,
(iv) Peroxy mano sulphuric acid,
(v) Peroxydithionic acid,
(vi) Dithionic acid,
(vii) Pyrosulphuric acid.
Answer:
Question 86.
Write the structure of the following:
- Sulphurous acid,
- Sulphuric acid,
- Thiosulphuric acid,
- Dithionous acid,
- Pyro sulphuric acid,
- peroxy mono sulphuric acid,
- Peroxy di sulphuric acid,
- Dithionic acid,
- Polythionic acid.
Answer:
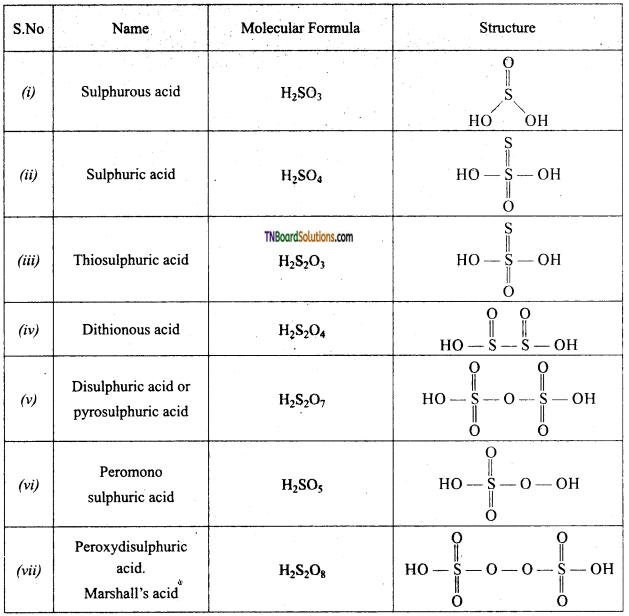
![]()
Question 87.
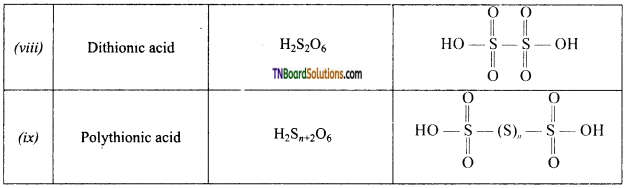
Briefly outline the trend in the physical properties of halogens.
Answer:
- Fluorine, chlorine, bromine, iodine and astatine constitute group 17 elements or halogens.
- The electronic configuration of these elements are
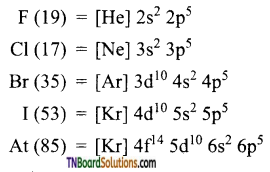
- Halogens have the smallest atomic radii in their respective periods. Both atomic and ionic radii increase from fluorine to iodine as the number of shells increases.
- All halogens exist as diatomic molecules in the elemental state. The different molecules of halogens are held together by vanderwaals forces of attraction. The strength of vanderwaals forces increases as. the size of the halogen atom increases from fluorine to iodine. As a result, F2 and Cl2 are gases, at room temperature, Br2 is a liquid, whereas I2 is a solid. Due to increase in vanderwaals force of attraction increases down the group, melting and boiling points increase with increase in atomic mass from fluorine to chlorine.
- All halogens are highly electronegative and electronegativity decreases down the group.
Question 88.
How is chlorine prepared from (i) NaCl (ii) HCl (iii) bleaching powder?
Answer:
(i) By the action of conc.H2SO4 in the presence of manganese dioxide an sodium chloride.
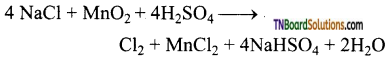
(ii) Oxidation of hydrochloric acid using oxidising agents such as MnO2, PbO2, KMnO4 or K2Cr2O7.

(iii) By treating to bleaching powder with a mineral acid.
![]()
Question 89.
Complete and balance the following equations.
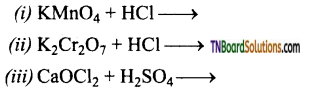
Answer:
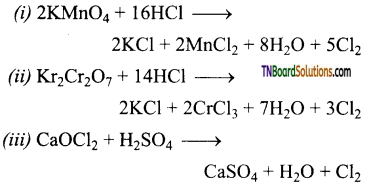
![]()
Question 90.
Briefly outline the manufacture of chlorine by
(i) Electrolytic process, (ii) Deacon’s process.
Answer:
(i) Electrolytic process: When a solution of brine (NaCl) is electrolysed, Na+ and Cl– ions are formed. Na+ ion reacts with OH– ions of water and forms sodium hydroxide. Hydrogen and chlorine are liberated as gases.
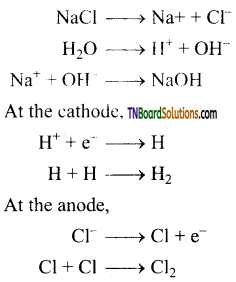
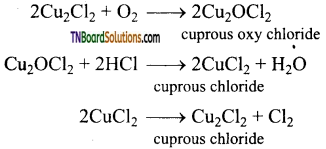
(ii) Deacon’s process: In this process, a mixture of air and hydrochloric acid is passed up a chamber containing a number of shelves, pumice stones soaked in cuprous chloride are placed. Hot gases at about 723 K are passed through a jacket that surrounds the chamber.
![]()
The chlorine obtained by this method is dilute and is employed for the manufacture of bleaching powder. The catalysed reaction is given below,
Question 91.
Give example for the reaction of chlorine an (i) Aluminium, (ii) Sulphur, (iii) boron, (iv) arsenic, (v) Phosphorus.
Answer:
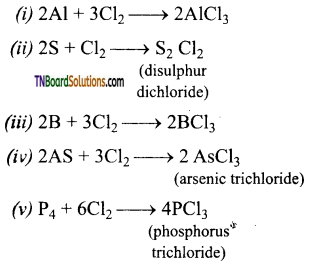
Question 92.
What happens when (i) chlorine is treated with excess ammonia (ii) ammonia is treated with an excess of chlorine? Give equation.
Answer:
(i) With excess ammonia, nitrogen gas is evolved.
![]()
(ii) With excess of chlorine, ammonium chloride is formed.
![]()
Question 93.
Give examples for the oxidising power of chlorine.
Answer:
Chlorine oxidises hydrogen sulphide to sulphur and liberates bromine and iodine from iodides and bromides. However, it doesn’t oxidise fluorides

Question 94.
What happens when chloride is treated with (i) cold, dilute sodium hydroxide and (ii) hot, concentrated solution of sodium hydroxide? Give equations.
Answer:
(i) When chlorine is treated with cold, dilute solution of sodium hydroxide, sodium chloride, sodium hypochlorite and water are formed.
![]()
(ii) When chlorine is treated with hot, concentrated solution of sodium hydroxide, sodium chloride, sodium chlorate and water are formed.
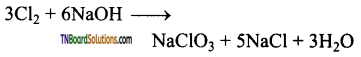
![]()
Question 95.
Explain the bleaching action of chlorine.
Answer:
Chlorine bleaches by oxidation

Colouring matter + Nascent oxygen → Colourless oxidation product The bleaching of chlorine is permanent.
Question 96.
Complete and balance the equation:
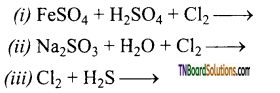
Answer:
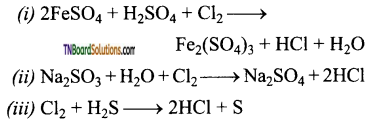
Question 97.
How is bleaching powder prepared? Give equation.
Answer:
Bleaching powder is produced by passing chlorine gas through dry slaked lime (calcium hydroxide).
![]()
Question 98.
Give a brief account of displacement reactions of halogens.
Answer:
In general, the halogen of lower atomic number will.displace the halide ion of higher atomic number.
Fluorine displaces other halogens from their corresponding halides.
![]()
chlorine displaces Br– and I– ions from their solutions and bromine displaces I– ion from their solutions.
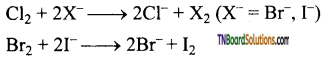
Question 99.
Mention the uses of chlorine.
Answer:
Uses of chlorine:
- Purification of drinking water.
- Bleaching of cotton textiles, paper and rayon.
- It is used in the extraction of gold and platinum.
Question 100.
How is hydrochloric acid prepared?
Answer:
Hydrochloric acid is prepared by heating sodium chloride with concentrated sulphuric acid and dissolving the hydrogen chloride formed in water.
![]()
![]()
Question 101.
How does hydrochloric acid react with (i) Zn, (ii) Na2CO3, (iii) Na2SO4? Give equation.
Answer:
(i) Hydrogen gas is liberated.
Zn + 2HCl → ZnCl2 + H2
(ii) Sodium carbonate is decomposed.
Na2CO3 + 2HCl → 2NaCl + H2O + CO2
(iii) Liberates sulphur dioxide from sodium sulphate.
Na2SO4 + 2HCl → 2NaCl + H2O + SO2.
Question 102.
What is aquaregia? Mention its uses.
Answer:
A mixture of concentrated hydrochloric acid and concentrated nitric acid in the ratio 3:1 is known as aquaregia. This is used for dissolving gold, platinum,
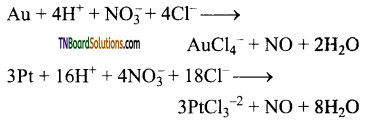
Question 103.
Mention the uses of hydrochloric acid.
Answer:
Uses of hydrochloric acid:
- Hydrochloric acid is used for the manufacture of chlorine, ammonium chloride, glucose from com starch etc.,
- Extraction of glue from bone and for purification of bone black.
Question 104.
Briefly outline the trend in physical and chemical properties of hydrogen halides.
Answer:
- At room temperature, hydrogen halides are gases but hydrogen fluoride can be readily liquefied. The gases are colourless but, with moist air gives white fumes due to the production of droplets of hydrohalic acid. In HF, due to the presence of strong hydrogen bond it has high melting and boiling points. This effect is absent in other hydrogen halides.
- Stability: The bond strength H—X decreases from HF to HI. Thus, HF is the most stable while HI is the least stable. The decrease is stability is due
to decrease in electronegativity from fluorine to iodine. This is reflected in the values of dissociation energy of H—X bond. - Volatility of the hydrides: A more volatile liquid must have a lower boiling point. The volatility of the hydrides shows the order HF < HI < HBr < HCl. The boiling point of HF is the highest due to extensive hydrogen bonding. As we move from HCl to HI, then boiling points show a regular increase due to a corresponding increase in the magnitude of vanderwaals force of attraction as the size of the halogen increases.
- Thermal stability: Thermal stability increases in the order HI< HBr < HCl < HF. Thermal stability is directly proportional to the bond dissociation energy. Since the bond dissociation energy of HF is the highest and that of HI is the least, therefore HF is the most stable halogen acid while the HI is the least stable halogen acid.
- Acid Strength: Aci(i strength increases in the order HF < HCl < HBr < HI. The strength of an acid depends upon its degree of ionisation which, in turn, depends upon the bond strength.

Thus higher the bond dissociation energy, lower is its degree of ionisation and weaker in the acid. Since bond dissociation energies of halogen acids increases in the order HI < HBr < HCl < HF the strength of acids increases in the reverse direction, i.e., HF < HCl < HBr < HI.
![]()
Question 105.
How are hydrogen fluoride and hydrogen chloride prepared from CaF2 and NaCl respectively? Give equation.
Answer:
HF and HCl are prepared by heating fluorides and chlorides, respectively with cone. H2SO4.

Question 106.
Hydrogen bromide and hydrogen iodide cannot be prepared by treating their bromides and iodides with cone. H2SO4. Why?
Answer:
HBr and HI are reducing agent and H2SO4 is an oxidising agent. The HBr or HI formed is oxidised to bromine or iodine.
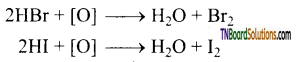
Question 107.
Give a method of preparation of hydrogen bromide and hydrogen iodide from sodium bromide and sodium iodide respectively.
Answer:
Phosphoric acid is used instead of cone H2SO4, because, conc H2SO4 oxidise HBr and HI formed to Br2 and I2 respectively.
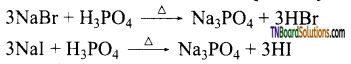
Question 108.
How is hydrogen bromide prepared from red phosphorus?
Answer:
To a paste of red phosphorus and water, bromine is added drop wise.
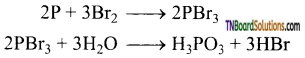
Question 109.
Give a method of preparation of hydrogen iodide from red phosphorus.
Answer:
By adding water drop wise to a mixture of red phosphorus and iodine.
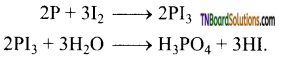
Question 110.
Hydrogen fluoride has a high melting and boiling point compared to other hydrogen halides. Give reason.
Answer:
In hydrogen fluoride, strong halogen bonding is present. This type of hydrogen bonding is not present in other halides. Hence, hydrogen fluoride has high melting or boiling point.
![]()
Question 111.
Fluorine forms hydrogen dihalides while other hydrogen halides do not form hydrogen dihalides. Give reason.
Answer:
HF is a stronger acid at high concentration
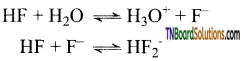
At high concentrations, the equilibrium involves the removal of fluoride ions is important, since, it affects the dissociation of hydrogen fluoride and increases the concentration of hydrogen ions. Hence, salts like NaHF2, KHF2, NH4HF2 are known. The other hydrogen halides do not form hydrogen dihalides.
Question 112.
Mention a reaction which is given by hydrogen fluoride alone, but not other hydrogen halides.
Answer:
Moist hydrofluoric acid (not dry) rapidly react with silica and glass.
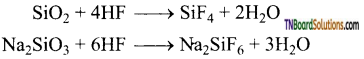
Question 113.
What is etching? Explain with an example.
Answer:
Glass being a mixture of sodium and calcium silicates react with hydro fluoric acid and forming sodium and calcium fluorosilicates respectively.
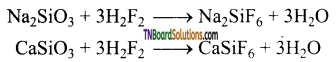
The etching of glass is based on these reactions. Thus, when hydrofluoric acid is added to glass, that portion is affected due to the formation of sodium and calcium fluorosilicates. This phenomenon is known as etching.
Question 114.
Compare to reducing power of the hydrogen halides.
Answer:
The reducing nature increases from HF to HI as the stability decreases from HF to HI. HF does not show any reducing nature. It cannot be oxidised by even by strong oxidising agents. HI is the strongest reducing agent.
It aqueous solution gets oxidised even by atmospheric oxygen.
The reducing action can be explained on the basis of increasing sizfe of the halide ion from F to I. The bigger ion can lose electron easily. HCl can be oxidised by strong oxidising agents like MnO2, KMnO4, K2Cr2O7
PbO2, Pb3O4 etc.
HBr acts as a strong reducing agent than HCl.
It can be oxidised by H2SO4 and atmospheric oxygen also

HI is the strongest reducing agent. It reduces H2SO4 to SO2, S, H2S, nitric acid to NO2, nitrous acid to NO, etc.
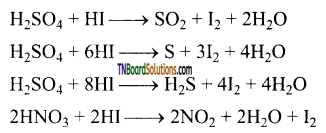
![]()
Question 115.
Mention the conditions for the formation of inter halogen compounds.
Answer:
- The central atom will be the larger one
- It can be formed only between two halogen and not more than two halogens.
Question 116.
Briefly outline the structure of interhalogens based on VSEPR theory,
(i) AB type,
(ii) AB3 type,
(iii) AB5 type,
(iv) AB7 type.
Answer:
(i) AB type: (eg: ClF, Br F, etc). In these molecules, the bigger atom undergoes sp3 hybridisation.
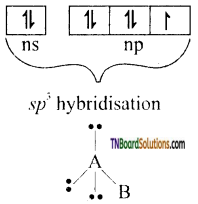
These hybrid orbitals contain one lone pair of electrons each,while the fourth orbital contain one electron. This hybrid orbital overlaps axially with the atomic orbital of B atom forming sigma bond. Thus theshape of the molecule is linear.
(ii) AB3 type: The central atom A undergoes sp3d hybridisation. The result is trigonal bi-pyramid structure.

Out of the five hybrid orbitals, two (equatorial) contain lone pair of electrons while the remaining three contain one electron each. The singly occupied hybrid orbitals overlap with the singly filled ‘p’ orbitals of an three B atoms forming three sigma bonds. The shape of the molecule AB3 is slightly bent T shaped with bond angles equal to 87.5°. eg: ClF3
(iii) Structure of AB5 type: The central atom A undergoes sp3d2 hybridisation.
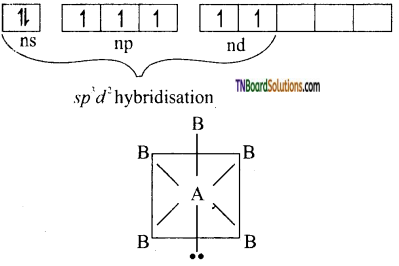
The result is an octahedral configuration one hybrid orbital contains a lone pair of electrons, while the remaining five have one electron each. Those singly occupied orbitals overlap with orbital of B atoms forming five sigma bonds. B atoms utilize their singly filled p orbital. The shape of the molecule is AB5 is square pyramidal. eg:BrF5
(iv) AB7 type: The central atom A undergoes sp3d3 hybridisation.
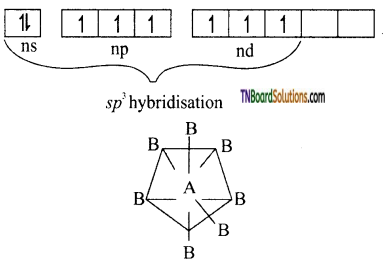
The structure is pentagonal bipyramid. Each of these seven hybrid orbitals is singly filled. Each of these hybrid orbitals overlaps with the singly filled ‘p’ orbitals of B atoms forming seven sigma bonds. Thus, the molecule, AB, has a pentagonal bi-pyramidal structure, eg: IF7
![]()
Question 117.
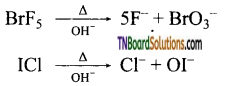
What happens when (i) BrF5 and (ii) ICl are treated with NaOH? Give equation.
Answer:
When heated with alkalies, the larger halogen form oxohalogen and the smaller forms halide ion.
Question 118.
Write a short note on oxides of halogens?
Answer:
- Fluorine form two oxygen compounds F2O and F2O2. These compounds are not called oxides but oxygen fluorides as fluorine is more electronegative than oxygen. The compounds of the rest of the halogens are termed as oxides.
- Halogens and oxygen do not combine readily with each other. The oxides of halogens are obtained indirectly.
- The important compounds of the halogens are listed below.

In these compounds, except fluorine (-1, oxidation state) all the other halogens have positive oxidation states. - Most of the halogen oxides are unstable and tend to explode when kept an standing or exposure to light.
- The iodine oxides are the most stable than chlorine oxides, but all the oxides of bromine decompose below the room temperature. The higher oxidation states are more to stable than the lower oxidation states.
- Oxygen fluorides do not form oxo acids. The oxides of other halogens are acidic. Acidic nature increases as the percentage of oxygen increases, i.e, Cl2O is less acidic than Cl2O7.
- All oxides are good oxidising agent. I2O5 is a very good oxidising agent.
Question 119.
Write a short note on oxy acids of halogens.
Answer:
Fluorine does not form any oxy acid as it is more electronegative than oxygen. Other halogens forms oxyacids of the type, HXO (hypohalous acid, +1 oxidation state), HXO2 (halous acid, + 3 oxidation state), HXO3 (halic acid, + 5 oxidation state, and HXO4 (perhalic acid, + 7 oxidation state. Some of these acids are unstable and are known 6nly in solutions or their salts.
Question 120.
Write a brief account of the trends in properties of group 18 (noble gases) elements.
Answer:
- Helium, neon, argon, krypton, xenon and Radan are the elements belong this group.
- Noble gases have the largest ionisation energy in a given period, as they have a completely filled orbital in their outer most shell. The first ionisation energy decreases from helium to radan.
- The electronic configuration of these elements are
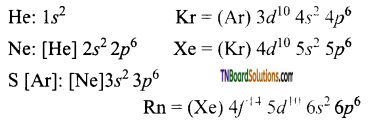
- Their atomic radius is known as vanderwaals radii. It is the highest in any period. The vanderwaals radii increases down the group.
- All have low values of melting and boiling points. These values increase gradually as atomic number increases. The low melting points is due to weak vanderwaals forces present between the atoms of the noble gases in liquid and solid states. These weak molecular forces, however, increase with increase in atomic size (mass) from He to Rn and therefore, the melting and boiling points increase gradually.
![]()
Question 121.
How are the following prepared? Give equations, (i) XeF2, (ii) XeF4, (iii) XeF6.
Answer:
Xenon fluorides are prepare by direct reaction of xenon and fluorine under different conditions as shown below.

Question 122.
Explain what happens when
(i) When XeF6 is heated at 50°C in a sealed quartz vessel.
(ii) When vapours of XeF6 is treated with water vapour.
(iii) When XeF6 reacts with 2.5 M sodium hydroxide.
Answer:
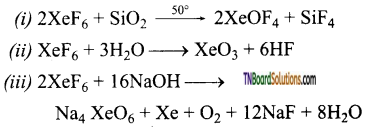
Question 123.
Give an example for the oxidising property of sodium perxenate.
Answer:
It oxidises Mn+2 ion to MnO4– ion as shown by the equation.
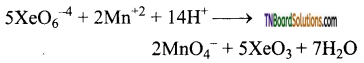
Question 124.
Name the addition compounds formed by the Xenon difluoride.
Answer:
Xenon difluoride forms addition compounds XeF2.2SbF5 and XeF2.2TaF5. Xenon hexa fluorides forms compound with boron and alkali metals. eg: XeF6.BF3, XeF6MF, M-alkali metals.
Question 125.
Explain the structure of Xenon fluoride (XeF2).
Answer:
- The Xenon in XeF undergoes sp3d hybridisation and the expected geometry is trigonal bi-pyramid. It has three lone pairs and two bond pairs.
- The Xenon and two fluorine atoms lie in a straight line while the three equatorial positions are occupied by lone pairs. The actual shape is linear.
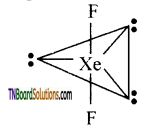
![]()
Question 126.
Explain the structure of Xenon tetrafluoride (XeF4).
Answer:
In the compound, Xenon is sp3d2 hybridised, i.e., The molecule is octahedral.
The Xenon and four fluorine atoms are coplanar, while two axial positions are occupied by the two lone pairs. The actual shape is square planar.
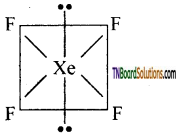
Question 127.
Explain the structure of Xenon hexa fluoride (XeF6).
Answer:
In the formation of XeF6, the XenOn atom undergoes sp3d3 hybridisation which gives the molecule is pentagonal bipyramid structure. Six positions are occupied by fluorine atoms and one position is occupied by lone pair of electrons. Due to the presence of lone pair of electron, the actual shape is distorted octahedral.
Question 128.
Explain the structure of Xenon oxy difluoride (XeOF2).
Answer:
It has a T shaped structure. Xenon undergoes sp3d3 hybridisation giving trigonal bipyramid configuration in which two position are occupied by lone pairs.
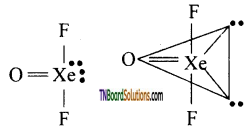
Question 129.
Explain the structure of Xenon oxy tetra fluoride (XeOF4).
Answer:
XeOF4 has a square pyramidal shape as shown below.
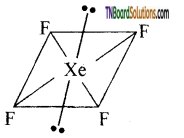
This shape results from sp2d3 hybridisation of Xenon atom. One position is occupied by a lone pair of electrons.
Question 130.
Explain the structure of Xenon trioxide (XeO3).
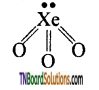
Answer:
The Xenon undergoes sp2 hybridisation. The structure is tetrahedral. Three oxygen atoms occupy three comers of a tetrahedral forming a sigma and a pi bond. Each with a hybrid and ‘d’ orbital of the xenon atom. The actual shape of the molecule is pyramidal as one position of a tetrahedron is occupied by lone pair.
Question 131.
Mention the uses of Xenon.
Answer:
- Xenon is used in fluorescent bulbs, flash bulbs and lasers.
- Xenon emits an intense light in discharge tubes instantly. Due to this it is used in high speed electronic flash bulbs used by photographers.
![]()
Question 132.
Mention the uses of radon.
Answer:
- Radon is radioactive and used as a source of gamma rays.
- Radon gas is sealed as small capsules and implanted in the body to destroy malignant i.e., cancer growth.
Choose the correct answer.
1. Which of the following is tribasic?
(a) H2PO2
(b) H3PO3
(c) H4P2O7
(d) H3PO4
Answer:
(d)
Hint: The structure of H3PO4 is
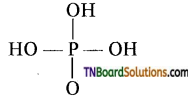
It has three replaceable hydrogen and hence it is tribasic.
2. Which of the following oxides of nitrogen is thermally most stable?
(a) N2O5
(b) NO2
(c) NO
(d) N2O
Answer:
(c)
3. P2O5 is extensively used as:
(a) reducing agent
(b) preservative
(c) oxidising agent
(d) dehydrating agent
Answer:
(d)
4. The range of oxidation states shown by phosphorus is from:
(a) – 3 to + 5
(b) – 3 to 0
(c) 0 to +5
(d) – 4 to +2
Answer:
(a)
![]()
5. The structural formula of hypo phosphorus acid is:
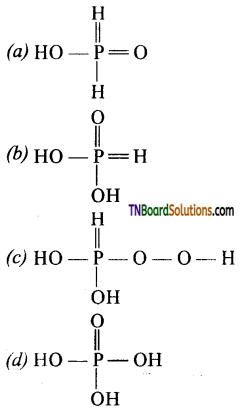
Answer:
(a)
6. Which of the following oxides is the most acidic?
(a) P2O5
(b) N2O5
(c) Sb2O3
(d) AS2O3
Answer:
(b)
Hint: Down the group acidic character decreases.
7. In white phosphorus molecule (P4) which one is not correct?
(a) Six P – P single bonds are present
(b) Four P – P single bonds are present
(c) Four lone pairs of electrons are present
(d) P – P – P bond angle is 60°
Answer:
(b)
8. In the preparation of sulphuric acid, V2O5 is used as a catalyst in the reaction?
(a) S + O2 → SO2
(b) 2SO2 + O2 → 2SO3
(c) SO3 + H2O → H2SO4
(d) none of these
Answer:
(b)
9. HCOOH reacts with conc. H2SO4 to produce:
(a) CO
(b) CO2
(c) SO2
(d) SO3
Answer:
(a)
10. The geometry of H2S and its dipole moment are:
(a) angular and non zero
(b) angular and zero
(c) linear and non zero
(d) linear and zero
Answer:
(a)
![]()
11. Among H2O, H2S, H2Te, H2Se, the one with maximum boiling point is:
(a) H20 because of hydrogen bonding
(b) H2Te because of higher molecular mass
(c) H2S because of hydrogen bonding
(d) H2Se because of lower molecular mass
Answer:
(a)
12. Which of the following has the highest bond energy?
(a) O – O
(b) S – S
(c) Se – Se
(d) Te – Te
Answer:
(a)
13. Which of the following reaction is not feasible?
(a) 2KI + Br2 → 2KBr + I2
(b) 2KBr + I2 → 2KI + Br2
(c) 2KBr + Cl2 → 2KCl + Br2
(d) 2H2O + F2 → 4HF + O2
Answer:
(b)
Hint: F2 can displace Cl2, Br2 and I2 from HCl, KBr and KI.
Cl2 can displace Br2 and I2 from KBr and KI.
Br2 can displace only I2 from KI.
I2 can displace none.
14. [X] + H2SO4 → [Y]. a colourless gas with initiating smell.
[Y] + K2Cr2O7 + H2SO4 → green solution
[X] and [Y] are:

Answer:
(a)
Hint: Sulphides on treatment with cone H2SO4, produce SO2 gas, which reduces acidified K2Cr2O7 to green Cr2 (SO4)3 solution.
15. Which of the following chemical reactions depicts the oxidising nature of conc. H2SO4?
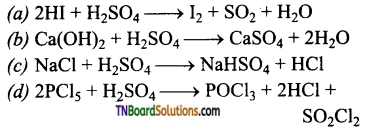
Answer:
(a)
![]()
16. The correct order of reactivity of halogens is:
(a) F > Br > Cl > I
(b) F > Cl > Br > I
(c) I > Br > Cl > F
(d) Br > Cl > F > I
Answer:
(b)
17. Which one of the following arrangements does not truly represent the property against it?
(a) Br2 < Cl2 < F2 Electronegativity
(b) Br2 < F2 < Cl2 Electron affinity
(c) Br2 < Cl2 < F2 Bond energy
(d) Br2 < Cl2 < F2 Oxidising power
Answer: (c)
Hint: F2 < Cl2 < Br2
18. Which products are expected from the disproportionation reaction of hypochlorus acid?
(a) HClO3 and Cl2O
(b) HClO2 and HClO3
(c) HCl and Cl2O
(d) HCl and HClO3
Answer: (d)
Hint- 3HOCl → 2HCl + HClO3
19. Among the halogens, the one which is oxidised by nitric acid is:
(a) F
(b) Cl
(c) Br
(d) I
Answer:
(d)
20. Sea divers go deep in sea water with a mixture of the following gases?
(a) O2 and Ar
(b) O2 and He
(c) CO2 and Ar
(d) O2 and CO2
Answer:
(b)
21. Among the following molecules:
(i) O2
(ii) XeOF4
(iii) XeF6
those having same number of lone pairs on Xe are:
(a) (i) and (ii) only
(b) (i) and (iii) only
(c) (ii) and (iii) only
(d) in all
Answer: (d)
![]()
22. As compared to nitrogen, oxygen is:
(a) less electronegative and less reactive
(b) more electronegative and less reactive
(c) more electronegative and more reactive
(d) less electronegative and more reactive
Answer:
(c)
23. Among the following the pair in which two species are not isostructural is:
(a) SiF4 and SF4
(b) IO3– and XeO3
(c) BH4– and NH4+
(d) PF6– and SF6
Answer:
(a)
Hint: SiF4 is tetrahedral while SF4 is square pyramidal.
24. Which one of the following statements is correct?
(a) The bond dissociation energy of fluorine is less than chlorine.
(b) Pene HBr can be prepared by treatment of NaBr with conc. H2SO4
(c) Hydrazine (N2H4) is a stronger base than NH3
(d) H2S is a weaker acid than H2O
Answer:
(c)
25. Match the compounds in list I with list II.
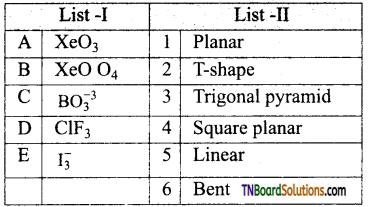
Code:
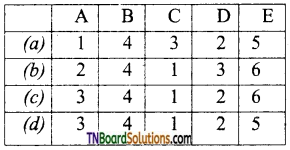
Answer:
(d)
26. Which of the following two are iso structural?
(a) XeF2, IF2–
(b) NH3, BF3
(c) CO3-2, SO3-2
(d) PCl5, ICl5
Answer:
(a)
![]()
27. Assertion (A): Nitrogen molecule is less reactive than molecular oxygen.
Reason (R): The bond length of N2 is shorter than that of oxygen.
(a) Both assertion and reason are true and reason is the correct explanation of assertion.
(b) Both assertion and reason are true, but reason is not the correct explanation of assertion.
(c) Assertion is true but reason is false.
(d) Both assertion and reason are false.
Answer:
(a)
28. Assertion (A): HClO4 is a stronger acid than HClO3.
Reason (R): The oxidation state of chlorine in HClO4 is +5 and in HClO3 is +7.
(a) Both assertion and reason are true and reason is the correct«explanation of assertion.
(b) Both assertion and reason are true, but reason is not the correct explanation of assertion.
(c) Assertion is true but reason is false.
(d) Both assertion and reason are false.
Answer:
(c)
Hint: The oxidation state of chlorine in HClO4 is +7 and that in HClO5 is +5.
29. Which reactions are used in the preparation of halogen acid?
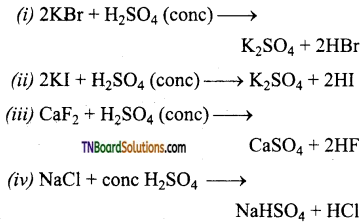
(a) (i) and (ii) only
(b) (ii) and (iii) only
(c) (iii) and (iv) only
(d) (i) and (iv) only
Answer:
(c)
30. Iodine cannot form the ion:
(a) I+2
(b) I–
(c) I+3
(d) I+
Answer:
(a)
![]()
31. Xenon forms compounds with fluorine under different conditions. The known fluorides are:
(i) XeF
(ii) XeF2
(iii) XeF3
(iv) XeF4
(a) (i) and (iv) only
(b) (ii) and (iv) only
(c) (ii) and (iii) only
(d) (i) and (iii) only
Answer:
(b)