Students get through the TN Board 12th Chemistry Important Questions Chapter 1 Metallurgy which is useful for their exam preparation.
TN State Board 12th Chemistry Important Questions Chapter 1 Metallurgy
Question 1.
Name a few metals that occur natively.
Answer:
Silver, gold, platinum, copper occur in nature in significant amounts.
Question 2.
Distinguish between ore and minerals.
Answer:
Both minerals and ores are naturally occurring compounds of the metal. It is possible to extract metal in large quantities and in an easy manner from ore but it is not so in a mineral. All ores are minerals but all minerals are not ores.
![]()
Question 3.
What do you understand by the term “concentration of the ore”?
Answer:
The ore particles are associated with earthy impurities like sand, clay etc. These impurities have to be removed before it is subjected to extraction. The removal of impurities from the ore by specific methods is known as the concentration of the ore.
Question 4.
Write the name and formula of the common ores of aluminium, iron and copper.
Answer:
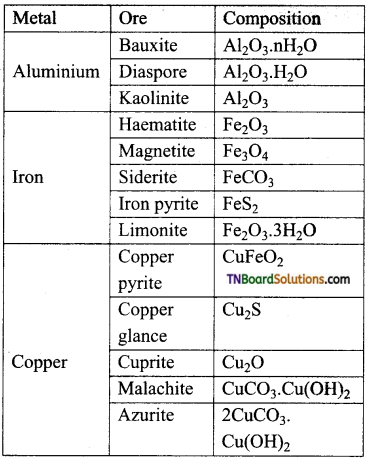
Question 5.
The names of a few ores are given. Write the composition of the ore. (i) Calamine, (ii) Zincite, (Hi) Galena, (iv) Cerrusite, (v) Cassiterite, (vi) Silver glance, (vii) Chloroargyrite.
Answer:
(i) Calamine: ore of Zn: ZnCO3
(ii) Zincite : ore of Zn: ZnO
(Hi) Galena: ore of Pb: PbS
(iv) Cerrusite: ore of Pb: PbCO3
(v) Cassiterite: ore of Sn: SnO2
(vi) Silver glance: ore of Ag: Ag2S
(vii) Chlorargyrite: ore of Ag: AgCl
![]()
Question 6.
Briefly give the principle involved in gravity separation of concentrating the ore.
Answer:
This method uses the difference in specific gravity between the ore particles and impurities (gangue). The crushed ore is treated with a rapidly flowing current of water. Lighter gangue particles are washed away, leaving behind heavier ore particles.
Question 7.
Why is the froth floatation process selected for the concentration of sulphide ores?
Answer:
The sulphide ore particles are preferentially wetted by oil, becomes lighter and thus rise to the surface along with the froth while the gangue particles are preferentially wetted by water, becomes heavier and settle down at the bottom of the tank. In this way, sulphide ore particles are separated and hence concentrated.
Question 8.
Name the chemicals used as frothing agents in the froth floatation process.
Answer:
Pine oil and eucalyptus oil are used as a frothing agent in the froth floatation process.
Question 9.
What is the role of depressant in the froth floatation process?
Answer:
The depressants help in the separation of two sulphide ores. For example, in the case of an ore containing PbS (galena) and ZnS (Zinc blende), the depressant is sodium cyanide (NaCN). It prevents ZnS from coming to the froth but allows PbS to come with froth in the froth floatation process ZnS forms a soluble complex with NaCN.
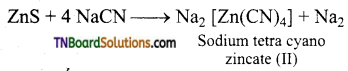
Question 10.
Explain the term leaching.
Answer:
It is a chemical separation of the ore and impurities. It involves the treatment of the ore with a suitable reagent to make it soluble while impurities remain insoluble. The insoluble impurities are separated by filtration.
![]()
Question 11.
Explain how, gold ore is concentrated by leaching, (or) How is leaching used to concentrate native ores of gold?
Answer:
The crushed ore of Au is leached with an aerated dilute solution of NaCN. Gold is converted into a soluble cyanide complex. The gangue, aluminosilicate remains insoluble.
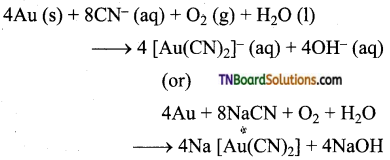
Question 12.
What is cementation? Explain with an equation.
Answer:
Gold can be recovered by reacting the deoxygenated leached solution with zinc. In this process, the gold is reduced to its elemental state (zero oxidation state) and the process is called cementation.
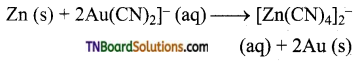
Question 13.
What is the role of ammonia in leaching ores containing nickel or copper or cobalt?
Answer:
When a crushed ore containing nickel, copper and cobalt is treated with aqueous ammonia under suitable pressure, ammonia selectively leaches these metals by forming their soluble complexes viz. [Ni(NH3)6]2+, [Cu(NH3)4]2+, and [CO(NH3)5H2O]3+ respectively from the ore leaving behind the gangue, iron (III) oxides/hydroxides and aluminosilicate.
Question 14.
Explain how bauxite ore is leached in the extraction of aluminium.
Answer:
The bauxite ore is treated with aqueous sodium hydroxide and heated to a temperature of around 470 – 520 K, at 35 atm. The aluminium dissolves forming a soluble complex, sodium meta aluminate leaving behind impurities.
Al2O3 (S) + 2NaOH (aq) + 3H2O (l) → 2Na [Al(OH)4] aq.
The hot solution is separated from its impurities and CO2 gas is passed. Pure alumina gas precipitated
2Na [Al(OH)4] (aq) + CO2 (g) → Al2O3. H20 + 2 NaHCO3 (aq)
The precipitate is washed and dried.
![]()
Question 15.
How is sulphide ores leached?
Answer:
The sulphide ores (ZnS, PbS) are treated with hot, aqueous sulphuric acid. The insoluble sulphide is converted to soluble sulphate and elemental sulphur which gets precipitated.
2ZnS (s) + 2H2SO4 (aq) + O2 (g) → 2ZnSO4 (aq) + 2S (s) + H2O
Question 16.
Explain the principle involved in the magnetic separation of the ore with an example.
Answer:
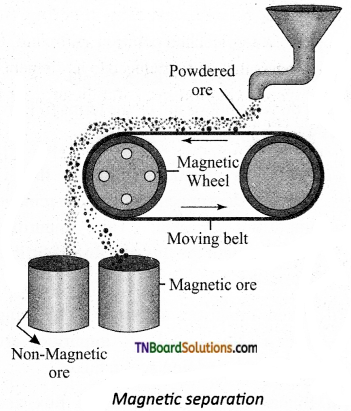
This method is based on the difference in magnetic properties of the ore and the impurities. For example, tinstone, an ore of tin can be separated from its magnetic impurities (wolframite). The crushed ore is poured onto an electromagnetic separator consisting of a belt moving over two rollers of which one is magnetic. The magnetic part of the ore is attracted towards the magnet and falls as a heap close to the magnetic region while the non-magnetic part falls away.
Question 17.
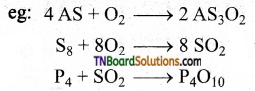
Give chemical equations involved in the process of roasting.
Answer:
Roasting converts sulphide ores to their oxides.

It also removes volatile impurities like moisture and non-metals as their oxides.
![]()
Question 18.
Explain the term ‘Roasting’ in metallurgy.
Answer:
Roasting is a process in which sulphide ores are converted to oxide ores by heating the ore in excess of oxygen in a reverberatory furnace below the melting point of the metal.
eg: Galena when roasted gets converted to lead monoxide.
2PbS + 3O2 → 2PbO + SO2
During roasting volatile and non-metallic impurities are removed. The ore is rendered porous so that further steps in the extraction process becomes easy.
Question 19.
What is calcination? Explain with an example.
Answer:
Calcination is the process in which the concentrated ore is heated strongly in the absence of air. During this process, the water of crystallisation present in hydrated oxides escapes as moisture; This method is carried out in a limited supply of air. For example, calcination of the carbonate ore, carbon dioxide example is expelled.
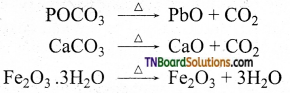
Question 20.
Distinguish between calcination and roasting.
Answer:
- Both processes are used to convert ores into metallic oxides. During roasting sulphide ores are converted to oxides and during calcination carbonate ores are converted to oxides.
- Roasting is carried out by heating the ore with excess oxygen below the melting point of the ore whereas calcination is done by heating the ore in a limited supply of oxygen.
- During roasting volatile impurities are removed and non-metallic impurities escape as their oxides. During calcination, the water of crystallisation escapes.
- Both processes render the ore porous which makes further steps of the extraction process easiest.
Question 21.
Write chemical equations involved in the calcination process?
Answer:
Calcination is the process that involves the conversion of carbonate ores to oxides.
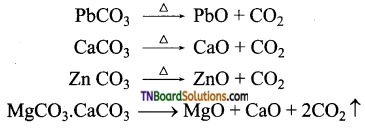
The water of crystallisation of hydrated salts escapes as water vapour.
Question 22.
Give a brief account of the reduction of metal oxides to metals.
Answer:
- Metal oxides can be reduced to metals by using a suitable reducing agent. The choice of the reducing agent depends on the nature of the metal. The reducing agents used commonly are carbon, carbon monoxide, hydrogen and reactive metals like sodium.
- Carbon is used as a reducing agent in the reduction of oxide ores such as ZnO, Mn3O4 and Cr2O3. The ore and coke are heated strongly in a blast furnace.

- Carbon monoxide is used to reduce ferric oxide in the extraction of iron.
Fe2O3 (s) + 3CO (g) → 2Fe (s) + 3CO2 (g) ↑ - Gaseous hydrogen is used to reduce oxides of less electropositive metals (Fe, Pb, Cu) than hydrogen.
Ag2O + H2 → 2Ag + H2O
Fe3O4 + 4H2 → 3Fe + 4H2O - Aluminium is used to reduce metal oxides like Cr2O3.
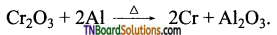
Active metals such as sodium, potassium and calcium are also used as reducing agents.
B2O3 + 6Na → 2B + 3Na2O
Rb2O3 + 3Mg → 2Rb + 3MgO
![]()
Question 23.
What do you understand by the terms (i) Flux, (ii) Slag? Explain with examples.
Answer:
- Flux is a chemical substance that is added to the concentrated ore, to remove impurities present. The impurities combine with the flux and form a fusible mass slag.
- Acidic impurities are removed by using a basic flux and basic impurities are removed by adding acidic fluxes.
- Silicon dioxide is an acidic flux and limestone (CaO) is a basic flux.
- CaO(s), the basic flux removes the gange silicon dioxide (SiO2), as calcium silicate (slag) in the extraction of iron.
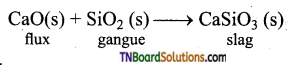
Slag: Slag is a compound formed when flux and gangue can combine with each other. The slag is a feasible mass, which removes, the impurities in the ore. In the extraction of iron, FeO is added as a flux to remove the gangue (SiO2) as ferrous silicate (slag).
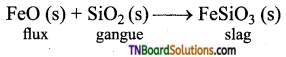
Question 24.
Give chemical equations involved in the extraction of copper from copper pyrites.
Answer:
- Smelting of the concentrated ore:
Cu2S and FeS formed in equation (1) is called mattle. - The remaining Cu2S is further oxidised to cuprous oxide, which is subsequently converted to metallic copper.
Cu2S + 3O2 → 2Cu2S + 2SO2
2Cu2O + Cu2S → 6Cu + SO2
Question 25.
Complete and balance the following equations.
Answer:
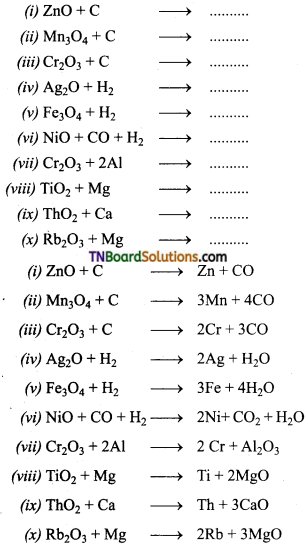
![]()
Question 26.
Briefly outline the thermodynamic principle involved in metallurgy.
Answer:
For any process, Gibb’s free energy change ΔG should be negative, i.e.,
ΔG = ΔH – TΔS should be negative where ΔH and ΔS are enthalpy and entropy changes respectively. The extraction of the metals from their oxides is carried out by using different reducing agents. A suitable reducing agent is chosen based on thermodynamics.
Let us consider the reduction of metallic oxide by carbon.
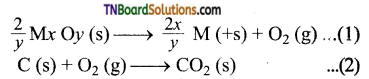
Thermodynamically, the reduction of metal oxide (equation -1), with a given reducing agent (equation -2) can occur if the free energy change for the coupled reaction is negative. Hence the reducing agent in chosen in such a way to produce a large negative value for the coupled reactions.
Question 27.
What is the Ellingham diagram? Explain.
Answer:
Ellingham’s diagram is used to obtain the ΔG° values at various temperatures for the reduction of metal oxides by treating reduction as an equilibrium process.
The diagram is made by plotting temperature in the x-axis and the standard free energy of formation (ΔG°) of the metal oxide in the ‘y ’ axis. The resultant plot is a straight line with ΔS as slope and ΔH as ‘y’ intercept. The graphical representation of the variation of standard free energy of formation of various metal oxides with temperature is known as the Ellingham diagram.
Question 28.
Briefly explain the salient features of the Ellingham diagram.
Answer:
(i) Ellingham diagram consists of plots that represent the variation of standard free energy of formation of metal oxides versus temperature.
(ii) These plots help is in predicting the feasibility of thermal reduction of ore. Consider the formation of metal oxide.
2xM (s) + O2 (g) → 2MxO (s)
In this reaction, there is a decrease in the value of ΔS° as M.rO is a solid and 02 is a gas. i.e., ΔS° is negative. Thus, if the temperature is increased, TΔS° becomes more negative. As in the equation ΔG° = ΔH° – TΔS°
TΔS° is substracted, therefore ΔG° becomes less negative i.e., ΔG° is likely to increase with temperature and this trend is confirmed from the curves of metal oxides.
The following observations are made from the curves.
(a) The slope of the curves of formation of metal oxides is positive because ΔG° becomes less negative and increases with temperature.
(b) Each curve is a straight line except when some change takes place in phase. The temperature at which such a change occur is indicated by an increase in slope on the positive side.
(c) In the case of CO, ΔG° decreases as ΔS° increases. This is indicated by the downward trend.
(d) Any metal oxide with lower ΔG° is more stable than a metal oxide with higher ΔG°. This implies that the metal oxide placed higher in the diagram can be reduced by the metal lower in the diagram.
Question 29.
Mention the limitations of the Ellingham diagram.
Answer:
- Ellingham diagram is constructed based only on thermodynamic considerations. It gives information about the thermodynamic feasibility of a reaction. It does not tell anything about the rate of the reaction. Moreover, it does not give any idea about the possibility of other reactions that might be taking place.
- The interpretation of ΔG is based on the assumption that the reactants are in equilibrium with the product which is not always true.
![]()
Question 30.
Based on the Ellingham diagram explain.
Answer:
- Aluminium can be used as a reducing agent for the reduction of chromic oxide.
In the Ellignhem diagram, the formation of chromium oxide lies above that of the aluminium, meaning that Al2O3 is more stable than Cr2O3. Hence aluminium can be used as a reducing agent for the reduction of chromic oxide. - Aluminium cannot be use4 to reduce MgO or CaO.
The formation of MgO or CaO lies below that, that of aluminium. Hence, it cannot be used to reduce oxides of Mg or Ca.
Question 31.
Why is the reduction of a metal oxide easier if the metal is in the liquid state at the temperature of reduction?
[OR]
Account for the fact that the reduction of a metal oxide is easier if the metal is formed in the liquid state at the temperature of reduction.
Answer:
Entropy is higher when the metal is in the liquid state than when it is in the solid-state. Therefore, the value of entropy change (ΔS°) of the reduction process is more positive, when the metal is formed in the liquid state. The value of TΔS° increases and that of ΔH° remains constant, the value of ΔG° becomes more negative and hence reduction becomes easier.
Question 32.
The choice of a reducing agent in a particular reduction depends on the thermodynamic factor. How far do you agree with this statement?
Answer:
Support your opinion with two examples.
From the Ellingham diagram, it is evident that any metal oxide with a lower AG° value is more stable than the metal oxide with a higher AG° value. This implies that the metal oxide placed lower in the diagram cannot be reduced by a metal involved in the formation of oxide placed higher in the diagram. However, the reverse can readily take place.
Thus, Al2O3 cannot be reduced by Cr however Cr2O3 can be reduced by Al.
At temperature 1733K, AG° formation of Al2O3 and Cr2O3 is given below.

The reduction of Al2O3 by Cr may be obtained by subtracting equation (i) and (ii).
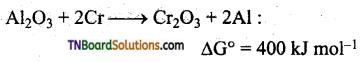
ΔG° = 400 kJ mol-1 This reaction is thermodynamically not feasible since ΔG° has a positive value. But the reverse reaction is possible as it has a negative ΔG° value. Similarly, both aluminium and zinc can reduce FeO to Fe, but Fe cannot reduce Al3O2 Or ZnO.
Question 33.
Based on the Ellingham diagram given below predict a suitable reducing agent for the reduction of Cu2O.
Answer:
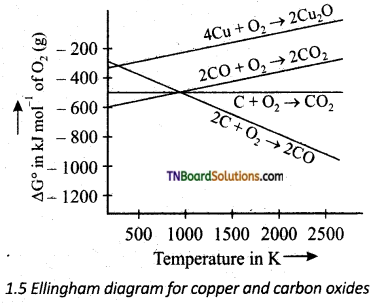
It is seen in the diagram that the Cu20 curve lies almost at the top while the lines sharing the formation of CO2 from C, and the formation of CO from carbon lie much below it. So it is easy to reduce Cu2O to the metal by heating it with coke at temperatures after 500 – 600 K.
Cu2O + C → 2Cu + CO
![]()
Question 34.
Show by using the Ellingham diagram given below to predict the possible temperature at which ZnO is reduced to Zn by carbon.
Answer:
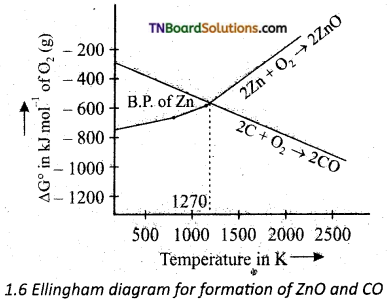
Ellingham diagram reveals that the lines involving the formation of ZnO and carbon monoxide cross each other at about 1270 K. There is a sudden increase in the value of ΔG° for the formation of ZnO above 1180°C. This is due to the fact that zinc begins to boil at this temperature. Above, 1270 K, ΔG° for the reaction.

is considered negative and thus ZnO is reduced by coke.
Question 35.
Out of carbon and carbon monoxide which is a better reducing agent for ZnO?
Answer:
From Ellingham’s diagram, it is clear that the free energy of formation of CO from C is lower at a temperature above 1120 K while that of CO2 from carbon is lower above 1323K than that of free energy of formation of ZnO. However, the free energy of the formation of CO2 from CO is always higher than that of ZnO. Hence, C is a better reducing agent of ZnO.
Question 36.
Explain why is the extraction of copper from pyrites difficult than that from its oxide ore through reduction.
Answer:
The standard free energy formation (ΔG°) for Cu2S is more negative than those of CS2 and H2S. Therefore neither carbon nor hydrogen can reduce Cu2S to copper. But the standard free energy of formation of Cu20 (ΔG°) is less negative than that of CO and hence carbon can readily reduce Cu2O to Cu.
Alternatively,
ΔG° / T line for CO has a negative slope and there is no compound CS analogous to CO with a steep negative ΔG°/T line. Thus, carbon is a good reducing agent for oxides and not for sulphides. Therefore sulphide ores are normally roasted in air to form oxides before reducing than with carbon.
Question 37.
Given the following ores indicate which of them Can be concentrated by magnetic separation method, (i) Bauxite, (ii) Kaolinite, (iii) Haematite, (iv) siderite, (v) iron pyrites, (vi) copper pyrites, (vii) galena, (viii) cassiterite, (ix) horn silver, (x) zincite, (xi) magnetite.
Answer:
Ores that are magnetic in nature can be separated from non-magnetic gangue particles by magnetic separation ores of iron such as Haematite (Fe2O3), Magnetite (Fe3O2), Siderite (FeCO3) and Iron pyrites (FeS2) being magnetic can be separated by magnetic separation.
![]()
Question 38.
Explain the principles involved in each of the following processes of refining metals. (i) Distillation, (ii) Liquation, (iii) Electrolyte refining, (iv) zone refining, (v) vapour phase method.
Answer:
- Distillation: This method is used for the purification of volatile metals like zinc, mercury, cadmium etc. The impure metal is heated in a retort and the vapours are condensed in separate receivers. The pure metal distils leaving behind the non-volatile having higher boiling point) impurities in the retort.
- Liquation: This method is used for the purification of metals in which the melting points of metals are lower than these of the impurities. The crude metal is heated in an inert atmosphere on a sloping hearth of a reverberatory furnace when the metal flows down into the receiver leaving behind infusible impurities on the hearth. Metals like tin and lead one purified by this method.
- Electrolyte refining:
Electrolytic refining is a process of purification of an impure metal by electrolysis. The impure metal is used as an anode and a pure metal is used as a cathode. The electrolyte is a suitable aqueous solution of the salt of the metal. During electrolysis, the metal dissolves at the anode, migrates to the cathode and gets deposited as a pure metal, eg: Electrolytic refining of silver:
Cathode: Pure silver Anode: Impure silver rods Electrolyte: Acidified aqueous solution of silver nitrate.
When a current is passed through the electrodes the following reactions will take place.
Reaction at anode 2Ag (s) → Ag+ (aq) + e–
Reaction at cathode Ag+ (aq) + e– → Ag(s)
During electrolysis, at the anode, the silver atoms lose electrons and enter the solution. The positively charged silver cations migrate towards the cathode and get discharged by gaining electrons and deposited on the cathode. Other metals such as copper, zinc etc., can also be refined by this process in a similar manner. - Zone refining: This method is based on the principles of fractional crystallisation. When an impure metal is melted and allowed to solidify, the impurities will prefer to be in the molten region, i.e., impurities are more soluble in the melt than in the solid-state metal. In this process, the impure metal is taken in the form of a rod. One end of the rod is heated using a mobile induction heater which results in the melting of the metal on that portion of the rod. When the heater is slowly moved to the other end the pure metal crystallises while the impurities will move on to the adjacent molten zone formed due to the movement of the heater. As the heater moves further away, the molten zone containing impurities also moves along with it. The process is repeated several times by moving the heater in the same direction again and again to achieve the desired purity level. This process is carried out in an inert gas atmosphere to prevent the oxidation of metals. Elements such as germanium (Ge), silicon (Si) and gallium (Ga) that are used as semiconductor are refined using this process.
(v) Vapour phase method:
The basic requirement is that the metal, when treated with a suitable reagent form a volatile compound which as decomposition gives the pure metal. eg: Nickel forms a volatile nickel carbonyl on heating with carbon monoxide, which on decomposition gives pure nickel.
Question 39.
Explain how zinc is purified by electrolytic refining.
Answer:
The impure zinc is made the anode while the cathode consists of pure sheets of zinc. The electrolyte is an acidified (H2SO4) solution of zinc sulphate (ZnSO4). On passing electric current pure zinc deposits on the cathode.
Question 40.
State the role of silver in the metallurgy of copper.
Answer:
During roasting, copper pyrites are converted to a mixture of FeO and Cu2O.
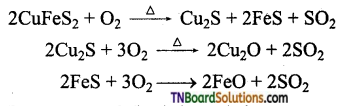
To remove FeO (basic impurity) an acidic flux, SiO2 is added during smelting. FeO combines with SiO2 to form ferrous silicate (FeSiO3) as slag and floats over the molten matter and hence can be easily removed.
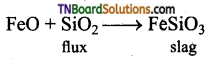
Question 41.
What is the role of graphite in the electrometallurgy of aluminium?
Answer:
In the electrometallurgy of aluminium, a fused mixture of alumina, cryolite and fluorspar (CaF2) is electrolysed using graphite as anode and graphite lined iron as a cathode. During electrolysis, Al is liberated at the cathode while CO and CO2 one liberated at the anode.
At Cathode: Al+3 (melt) → Al (l)
At anode: C + O-2 (melt) → CO + 2e–
C + 2O-2 (melt) → CO2 + 4e–
Instead of graphite, some metal is used as an anode, then O2 liberated will not only oxidise the metal of the electrode but would also convert the same Al liberated at the cathode back to Al2O3.
Since graphite is much cheaper than any metal, it is used as a cathode. Thus the role of graphite in the electrometallurgy of Al is to prevent the liberation of 02 at the anode. Which may otherwise convert Al to Al2O3.
![]()
Question 42.
Mention the application of the following metals, (i) Al, (ii) Cu, (iii) Fe, (iv) Au.
Answer:
Al: Aluminium is the most abundant metal and is a good conductor of electricity and heat. It also resists corrosion. The following are some of its applications.
- Many heat exchangers/sinks and our day to day cooking vessels are made of aluminium.
- It is used as wraps (aluminium foils) and is used in packing materials for food items.
- Aluminium is not very strong, However, its alloys with copper, manganese, magnesium and silicon are lightweight and strong and they are used in the design of aeroplanes and other forms of transport.
- As aluminium shows high resistance to corrosion, it is used in the design of chemical reactors, medical equipment, refrigeration units and gas pipelines.
- Aluminium is a good electrical conductor and cheap, hence used in electrical overhead electric cables with a steel core for strength.
Cu:
- Copper is used for making coins and ornaments along with gold and other metals.
- Copper and its alloys are used for making wires, water pipes and other electrical parts.
Fe:
- Iron and its alloys are used everywhere including bridges, electricity pylons, bicycle chains, cutting tools and rifle barrels.
- Cast iron is used to make pipes, valves and pumps stoves etc…
- Magnets can be made of iron and its alloys and compounds.
- An important alloy of iron is stainless steel, and it is resistant to corrosion. It is used in architecture, bearings, cutlery, surgical instruments and jewellery. Nickel steel is used for making cables, automobiles and aeroplane parts. Chrome steels are used for manufacturing cutting tools and crushing machines.
Au:
- Gold is used for coinage and has been used as the standard for monetary systems in some countries.
- It is used extensively in jewellery in its alloy form with copper. It is also used in electroplating to cover other metals with a thin layer of gold which are used in watches, artificial limb joints, cheap jewellery, dental fillings and electrical connectors.
- Gold nanoparticles are also used for increasing the efficiency of solar cells and also used as catalysts.