Tamilnadu State Board New Syllabus Samacheer Kalvi 12th Chemistry Guide Pdf Chapter 7 Chemical Kinetics Text Book Back Questions and Answers, Notes.
Tamilnadu Samacheer Kalvi 12th Chemistry Solutions Chapter 7 Chemical Kinetics
12th Chemistry Guide Chemical Kinetics Text Book Questions and Answers
Part – I Text Book Evaluation
I. Choose the correct answer
Question 1.
For a first order reaction A → B the rate constant is x min-1. If the initial concentration of A is 0.01 M, the concentration of A after one hour is given by the expression.
(a) 0.01 e-x
(b) 1 × 10-2 (1 – e-60x)
(c) (1 × 10-2) e-60x
(d) none of these
Answer:
(c) (1 × 10-2) e-60x
Solutions:
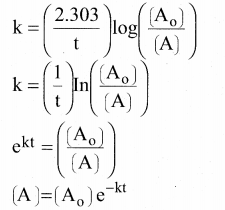
In this case
k = x min-1 and
[A0] = 0.01 M = 1 × 10-2 M
t = 1 hour = 60 min
[A] = 1 × 10-2(e-60x)
![]()
Question 2.
A zero order reaction X → Product. with an initial concentration 0.02M has a half life of 10 min. If one starts with concentration 0.04M, then the half life is …………….
(a) 10 s
(b) 5 min
(c) 20 min
(d) cannot be predicted using the given information
Answer:
(c) 20 min
Solutions:
Given,
[A0] = 0.02 M ; t1/2 = 10 min
[A0] = 0.04 M ; t1/2 = ?
Substitute in (1)
10 min ∝ 0.02 M ……………………..(2)
t1/2 ∝ 0.04 M ……………………..(3)
Dividing Eq.(3) by Eq. (2) we get,
\(\frac { { t }^{ 1/2 } }{ 10min }\) = \(\frac { 0.04M }{ 0.02M }\)
t1/2 = 2 × 10 min = 20 min
Question 3.
Among the following graphs showing variation of rate constant with temperature (T) for a reaction, the one that exhibits Arrhenius behavior over the entire temperature range is ……………
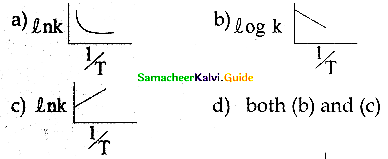
Answer:
b
Solution:
\(\text { Ae }\left(\frac{\mathrm{Ea}}{\mathrm{RT}}\right)\)
In k = In A – \(\left( \frac { { E }_{ a } }{ R } \right)\) \((\frac { 1 }{ T })\)
this equation is in the form of a straight line equation
y = c + m x
a plot of ink vs \((\frac { 1 }{ T })\) is a straight line with negative slope.
![]()
Question 4.
For a first order react ion A → product with initial concentration x mol L-1, has a half life period of 2.5 hours. For the same reaction with initial concentration mol L-1 the half life is
(a) (2.5 × 2) hours
(b) \((\frac { 2.5 }{ 2 })\) hours
(c) 2.5 hours
(d) Without knowing the rate constant, t1/2 cannot be determined from the given data
Answer:
(c) 2.5 hours
Solutions:
For a first order reaction
t1/2 = \(\frac { 0.693 }{ k }\) t1/2 does not depend on the initial concentration and it remains constant (whatever may be the initial concentration)
t1/2 = 2.5 hrs .
Question 5.
For the reaction, 2NH3 → N2 + 3H2, if
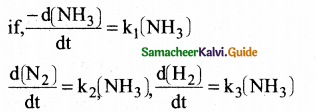
then the relation between
k1, k2 and k3 is
(a) k1 = k2 = k3
(b) k1 = 3k2 = 2k3
(c) 1.5k1 = 3k2 = k3
(d) 2k1 = k2 = 3k3
Answer:
(c) 1.5k1 = 3 k2 = k3
Solution:
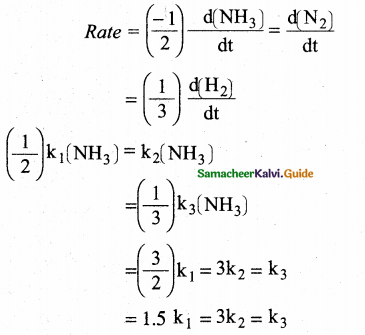
![]()
Question 6.
The decomposition of phosphine (PH3) on tungsten at low pressure is a first order reaction. It is because the …………….
(a) rate is proportional to the surface coverage
(b) rate is inversely proportional to the surface coverage
(c) rate is independent of the surface coverage
(d) rate of decomposition is slow
Answer:
(c) rate is independent of the surface coverage
Solution:
Given:
At low pressure, the reaction follows first-order, therefore
Rate ∝ [reactant]1
Rate ∝ (surface area)
At high pressure due to the complete coverage of the surface area, the reaction follows zero-order.
Rate ∝ [reactant]°.
Therefore the rate is independent of surface area.
Question 7.
For a reaction Rate = k [acetone]3/2 then a unit of the rate constant and rate of reaction respectively is …………..
(a) (mol L-1 s-1), (mol-1/2 L1/2 s-1)
(b) (mol-1/2 L1/2 s-1), (mol L-1 s-1)
(c) (mol1/2 L1/2 s-1), (mol L-1 s-1)
(d) (mol L s-1), (mol1/2 L1/2 s)
Answer:
(b) (mol1/2 L1/2 s-1), (mol L-1 s-1)
Solution:
Rate = k [A]n
Rate = \(\frac { -d[A] }{ dt } \)
unit of rate = \(\frac { mol{ L }^{ -1 } }{ s }\) = mol L-1 s-1
unit of rate constant = \(\frac { (mol{ L }^{ -1 }{ S }^{ -1 }) }{ { (mol{ L }^{ -1 }) }^{ n } }\)
= mol1-n Ln-1 S-1
rate = k [Acetone]3/2
n = 3/2
mol1-(3/2) L(3/2)-1 s-1
mol-(1/2) L(1/2) s-1
Question 8.
The addition of a catalyst during a chemical reaction alters which of the following quantities?
(a) Enthalpy
(b) Activation energy
(c) Entropy
(d) Internal energy
Answer:
(b) Activation energy
Solution:
Activation energy:
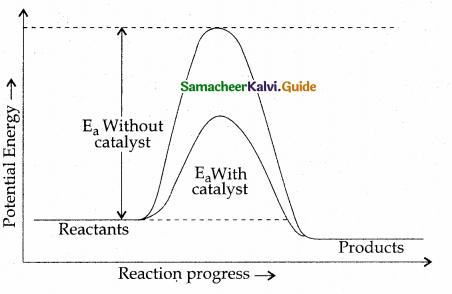
A catalyst provides a new path to the reaction with low activation energy. i.e, it lowers the activation energy.
![]()
Question 9.
Consider the following statements:
(i) increase in the concentration of the reactant increases the rate of a zero-order reaction.
(ii) rate constant k is equal to collision frequency A if Ea = ∞
(iii) rate constant k is equal to collision frequency A if Ea = o
(iv) a plot of ln (k) vs T is a straight line.
(v) a plot of ln (k) vs \((\frac { 1 }{ T })\) is a straight line with a positive slope.
Correct statements are
(a) (ii) only
(b) (ii) and (iv)
(c) (ii) and (v)
(d) (i), (ii) and (v)
Answer:
(a) (ii) only
Solutions:
In zero order reactions, an increase in the concentration of reactant does not alter the rate, So statement (i) is wrong.
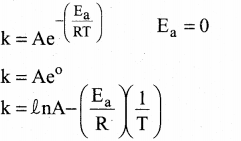
this equation is in the form of a straight line equation yc + mx. a plot of Ink vs \(\frac { 1 }{ T }\) is a straight line with negative slope so statements (iv) and (v) are wrong.
![]()
Question 10.
In a reversible reaction, the enthalpy change and the activation energy in the forward direction are respectively – x kJ mol-1 and y kJ mol-1. Therefore, the energy of activation in the backward direction is ………..
(a) (v – x) kJ mol-1
(b) (x + y) J mol-1
(c) (x – y) kJ mol-1
(d) (x + y) × 103 J mol-1
Answer:
(d) (x + y) × 103 J mol-1
Solution:
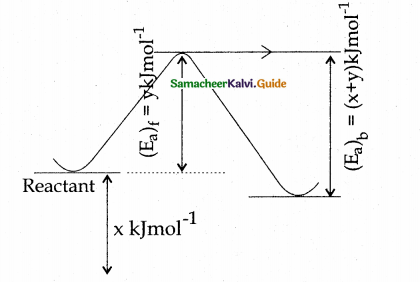
(x + y)kJmol-1
(x + y) × 103Jmol-1
![]()
Question 11.
What is the activation energy for a reaction if its rate doubles when the temperature is raised from 200K to 400K? (R 8.314 JK-1 mol-1)
(a) 234.65 kJ mol-1
(b) 434.65 kJ mol-1
(c) 2.305 KJ mol-1
(d) 334.65 J mol-1
Answer:
(c) 2.305 KJ mol-1
Solutions:


Question 12.
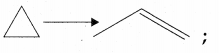
This reaction follows first order kinetics. The rate constant at particular temperature is 2.303 × 10-2 hour-1. The initial concentration of cyclopropane is 0.25 M. What will be the concentration of cyclopropane after 1806 minutes? (Log 2 = 0.30 10)
(a) 0.125 M
(b) 0.215 M
(c) 0.25 × 2.303 M
(d) 0.05 M
Answer:
(b) 0.2 15 M
Solution:

![]()
Question 13.
For a first-order reaction, the rate constant is 6.909 min-1. The time taken for 75% conversion in minutes is …………
(a) \((\frac { 3 }{ 2 })\) log 2
(b) \((\frac { 3 }{ 2 })\) log 2
(c) \((\frac { 3 }{ 2 })\) log \((\frac { 3 }{ 4 })\)
(d) \((\frac { 2 }{ 3 })\) log \((\frac { 4 }{ 3 })\)
Answer:
(b) \((\frac { 3 }{ 2 })\) log 2
Solution:
k = \((\frac { 2.303 }{ t })\) log \(\left( \frac { \left[ { A }_{ 0 } \right] }{ \left[ A \right] } \right)\)
[A0]= 100; [A]=25
6.909 = \((\frac { 2.303 }{ t })\) log \((\frac { 100 }{ 25 })\)
t = \((\frac { 2.303 }{ 6.909 })\) log (4)
⇒ t = \((\frac { 1 }{ 3 })\) log 22
t = \((\frac { 2 }{ 3 })\) log 2
Question 14.
In a first-order reaction x → y; if k is the rate constant and the initial concentration of the reactant x is 0.1 M, then, the half-life is ……..
(a) \((\frac { log2 }{ k })\)
(b) \((\frac { 0.693 }{ (0.1)k })\)
(c) \((\frac { In2 }{ k })\)
(d) none of these
Answer:
(c) \((\frac { In2 }{ k })\)
Solution:
k = \((\frac { 1 }{ t })\) In \(\left( \frac { \left[ { A }_{ 0 } \right] }{ \left[ A \right] } \right)\)
[A0] = 0.1
[A] = 0.05
k = \(\left( \frac { 1 }{ { t }_{ 1/2 } } \right)\) In \((\frac { 0.1 }{ 0.05 })\)
k = \(\left( \frac { 1 }{ { t }_{ 1/2 } } \right)\) In (2) ⇒ t1/2 = \((\frac { In(2) }{ k })\)
![]()
Question 15.
Predict the rate law of the following reaction based on the data given below:
2A + B → C + 3D
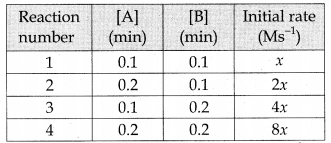
(a) rate = k [A]2 [B]
(b) rate = k [A][B]2
(c) rate = k [A][B]
(d) rate = k [A]1/2 [B]3/2
Answer:
(b) rate = k [A][B]2
Solution:
rate1 = k [0.1]n [0.1]m ……………(1)
rate2 = k [0.2]n [0.1]m …………(2)
Dividing Eq.(2) by Eq.(1)
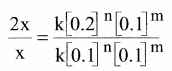
\(\frac { 2x }{ x }\) = 2n
∴ n = 1
rate3 = k [0.1]n [0.2]m …………..(3)
rate4 = k [0.2]n [0.2]m …………..(4)
Dividing Eq.(4) by Eq.(2)
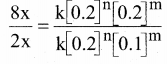
\(\frac { 8 }{ 2 } \) = 2m
∴ m = 2
∴ rate = k [A]1 [B]2
Question 16.
Assertion: rate of reaction doubles when the concentration of the reactant is doubled if it is a first-order reaction.
Reason: rate constant also doubles
(a) Both assertion and reason are true and reason is the correct explanation of assertion.
(b) Both assertion and reason are true but reason is not the correct explanation of assertion.
(c) Assertion is true but reason is false.
(d) Both assertion and reason are false.
Answer:
(c) Assertion is true but the reason is false.
Solution:
For a first reaction, when the concentration of reactant is doubled, then the rate of reaction also doubled. Rate constant is independent of concentration and is a constant at a constant temperature, i.e., it depends on the temperature, and hence, it will not be doubled and when the concentration of the reactant is doubled.
![]()
Question 17.
The rate constant of a reaction is 5.8 × 10-2 s-1. The order of the reaction is ………….
(a) First order
(b) zero-order
(c) Second-order
(a) Third order
Answer:
(a) First order
Solution:
The unit of the rate constant is s-1 and it indicates that the reaction is first order.
Question 18.
For the reaction N2 O5(g) → 2NO2(g) +\(\frac { 1 }{ 2 }\) O2(g) the value of rate of disappearance of N2O5 is given as 6.5 × 10-2 mol L-1s-1 The rate of formation of NO2 and O2 is given respectively as
(a) (3.25 × 10-2 mol L-1s-1) and (1.3 × 10-2 mol L-1s-1)
(b) (1.3 × 10-2 mol L-1s-1) and (3.25 × 102 mol L-1s-1)
(c) (1.3 × 10-1 mol L-1s-1) and (3.25 × 10-2 mol L-1s-1)
(d) None of these
Answer:
(c) (1.3 × 10-1 mol L-1s-1) and (3.25 × 10-2 mol L-1s-1)
Solution:
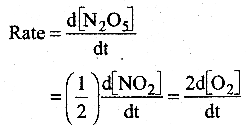
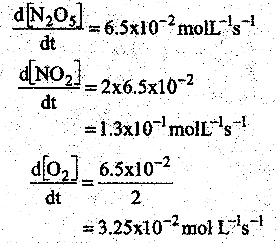
Question 19.
During the decomposition of H2O2 to give dioxygen, 48g O2 is formed per minute at a certain point in time. The rate of formation of water at this point is …………….
(a) 0.75 mol min-1
(b) 1.5 mol min-1
(c) 2.25 mol min-1
(d) 3.0 mol min-1
Answer:
(d) 3.0 mol min-1
Solution:
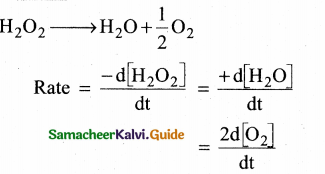
No. of moles of oxygen = \((\frac { 48 }{ 32 })\) = 1.5 mol
Rate of formation of oxygen = 2 × 1.5 = 3 mol min-1
![]()
Question 20.
If the initial concentration of the reactant is doubled, the time for half-reaction is also doubled. Then the order of the reaction is …………
(a) Zero
(b) one
(c) Fraction
(d) none
Answer:
(a) Zero
Solution:
For a first order reaction t1/2 is independent of initial concentration .i.e., n \(\neq\) 1 for such cases
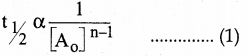

Question 21.
In a homogeneous reaction A → B + C + D, the initial pressure was P0 and after time t it was P. Expression for rate constant in terms of P0, P and t will be

Answer:
(a)
Solution:


![]()
Question 22.
If 75% of a first-order reaction was completed in 60 minutes, 50% of the same reaction under the same conditions would be completed in ………
(a) 20 minutes
(b) 30 minutes
(c) 35 minutes
(d) 75 minutes
Answer:
(b) 30 minutes
Solution:
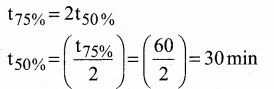
Question 23.
The half-life period of a radioactive element is 140 days. After 560 days, 1 g of the element will be reduced to
(a) \(\frac { 1 }{ 2 }\) g
(b) \(\frac { 1 }{ 4 }\) g
(c) \(\frac { 1 }{ 8 }\) g
(d) \(\frac { 1 }{ 16 }\) g
Answer:
(d) \(\frac { 1 }{ 16 }\) g
Solution:
in 140 days ⇒ initial concentration reduced to \(\frac { 1 }{ 2 }\) g
in 280 days ⇒ initial concentration reduced to \(\frac { 1 }{ 4 }\) g
in 420 days ⇒ initial concentration reduced to \(\frac { 1 }{ 8 }\) g
in 560 days ⇒ initial concentration reduced to \(\frac { 1 }{ 8 }\) g
Question 24.
The correct difference between first and second-order reactions is that …………
(a) A first-order reaction can be catalysed a second-order reaction cannot be catalysed.
(b) The half-life of a first-order reaction does not depend on [A0] the half-life of a second-order reaction does depend on [A0].
(c) The rate of a first-order reaction does not depend on reactant concentrations; the rate of a second-order reaction does depend on reactant concentrations.
(d) The rate of a first-order reaction does depend on reactant concentrations; the rate of a second-order reaction does not depend on reactant concentrations,
Answer:
(b) The half-life of a first-order reaction does not depend on [A0]; the half-life of a second-order reaction does depend on [A0].
Solution:
For a first order reaction
t1/2 = \(\frac { 0.6932 }{ k }\)
For a second order reaction
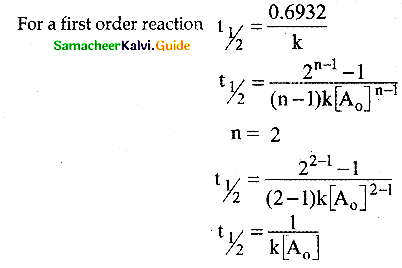
![]()
Question 25.
After 2 hours, a radioactive substance becomes \((\frac { 1 }{ 16 })\)th of original amount. Then the half life (in mm) is ………………
(a) 60 minutes
(b) 120 minutes
(c) 30 minutes
(d) 15 minutes
Answer:
(c) 30 minutes
Solution:
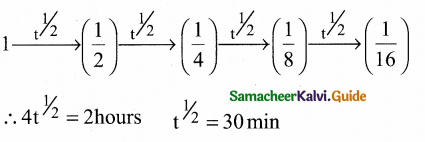
II. Answer the following questions:
Question 1.
Define average rate and instantaneous rate.
Answer:
1. Average rate:
The average rate of a reaction is defined as the rate of change of concentration of a reactant (or of a product) over a specified measurable period of time.
2. instantaneous rate:
Instantaneous rate of reaction gives the tendency of the reaction at a particular point of time during its course (or) The time derivative of the concentration of a reactant (or product) converted to a positive number is called the instantaneous rate of reaction.
Question 2.
Define rate law and rate constant.
Answer:
1. Rate law: The expression in which reaction rate is given in terms of molar concentration of the reactants with each term raised to some power, which may or may not be same as the Stoichiometric coefficient of the reacting species in a balanced chemical equation.
x A + y B → products
Rate = k [A]m [B]m
k = Rate constant
Rate constant is the proportionality constant equal to the rate of the reaction when the concentration of each reactant is unity.
In above rate law if [A] [B] = 1, rate constant k = Rate .
Question 3.
Derive integrated rate law for a zero-order reaction A → product.
Answer:
A reaction in which the rate is independent of the concentration of the reactant over a wide range of concentrations is called zero-order reactions. Such reactions are rare. Let us consider the following hypothetical zero-order reaction.
A → Product
The rate law can be written
Rate = k [A]°
(∴[A]° = 1)
– d [A] = k (1)
\(\frac { -d[A] }{ dt }\) = k(1)
-d[A] = k dt
Integrate the above equation between the limits of [A0] at zero time and [A] at some later time ‘t’,

![]()
Question 4.
Define the half-life of a reaction. Show that for a first-order reaction half-life is independent of initial concentration.
Answer:
The half-life of a reaction is defined as the time required for the reactant concentration to reach one half of its initial value.
For a first-order reaction, the half-life is a constant i.e., it does not depend on the initial concentration. The rate constant for a first-order reaction is given by,
For a first-order reaction

Question 5.
What is an elementary reaction? Give the differences between the order and molecularity of a reaction.
Answer:
Elementary reaction – Each and every single step in a reaction mechanism is called an elementary reaction. Differences between order and molecularity:
Order of a reaction:
- It is the sum of the powers of concentration terms involved in the experimentally determined rate law.
- It can be zero (or) fractional (or) integer.
- It is assigned for an overall reaction.
Molecularity of a reaction:
- It is the total number of reactant species that are involved in an elementary step.
- It is always a whole number, cannot be zero or a fractional number.
- It is assigned for each elementary step of the mechanism.
![]()
Question 6.
Explain the rate-determining step with an example.
Answer:
Consider the decomposition of hydrogen peroxide catalysed by I–
2H2O2(aq) → 2H2O(l) + O2(g)
Experimentally it is found that the reaction is first order with respect to both H2O2 and I–, which indicates that I– is also involved in the reaction.
The mechanism involves two steps.
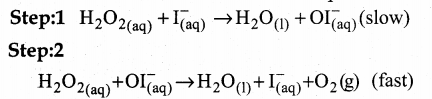
These two are elementary reactions. Adding step 1 and step 2 gives the overall reaction.
Step 1: is the rate-determining slow step, since it involves both H2O2 and I. The overall reaction is bimolecular.
Question 7.
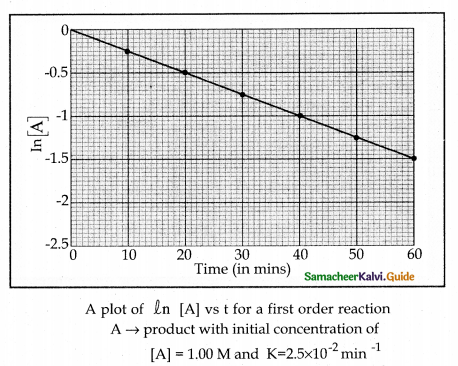
Describe the graphical representation of first order reaction.
Answer:
Rate constant for first order reaction is,
kt = ln\(\left( \frac { \left[ { A }_{ 0 } \right] }{ \left[ A \right] } \right)\)
kt = In [A0] – In [A]
In[A] = In [A0] – kty
y = c + mx
If we follow the reaction by measuring the concentration of the reactants at regular time interval ‘t’, a plot of ln[A] against ‘t’ yields a straight line with a negative slope. From this, the rate constant is calculated.
![]()
Question 8.
Write the rate law for the following reactions.
a) A reaction that is 3/2 order in x and zero-order in y.
b) A reaction that is second order in NO and first order in Br2.
Answer:
Rate =k[x]3/2[y]o
b) Rate = k[NO]2[Br2]1
Rate = k[x]3/2
Question 9.
Explain the effect of a catalyst on reaction rate with an example.
Answer:
- Significant changes in the reaction can be brought out by the addition of a substance called a catalyst.
- A catalyst is a substance which alters the rate of a reaction without itself undergoing any permanent chemical change.
- They may participate in the reaction, but again regenerate and the end of the reaction.
- In the presence of a catalyst, the energy of activation is lowered and hence, the greater number of molecules can cross the energy barrier and change over to products, thereby increasing the rate of the reaction.
- For example, the decomposition of potassium chlorate is enhanced by the addition of MnO2.
Question 10.
The rate law for a reaction of A, B, and C have been found to be rate = k[A]2 [B][L]3/2. How would the rate of reaction change when
- The concentration of [L] is quadrupled
- The concentration of both [A] and [B] are doubled
- The concentration of [A] is halved
- The concentration of [A] is reduced to(1/3) and the concentration of [L] is quadrupled.
Solution:
Rate = k [A]2 [B] [L]3/2 ………….(1)
1. when [L] = [4L]
Rate = k [A]2 [B] [4L]3/2
Rate = 8 (k[A]2 [B] [L]3/2) …………………..(2)
Comparing (1) and (3) rate is increased by 8 times.
2. when [A] = [2A] and [B] = [2B]
Rate = k[2A]2 [2B ] [L]3/2
Rate = 8 (k[A]2 [B] [L]3/2 …………….(3)
Comparing (1) and (3); rate is increased by 8 times.
3. when [A] = \([\frac { A }{ 2 }]\)
Rate = k \([\frac { A }{ 2 }]\)2 [L]\(\frac { 3 }{ 2 }\)
Rate = \(\frac { 1 }{ 4 }\) (k[A]2 [B] [L]3/2) ……………..(4)
Comparing (1) and ( 4); rate is reduced to \(\frac { 1 }{ 4 }\) times.
4. when [A] = \([\frac { A }{ 3 }]\) and [L] = [4L]
Rate k\(\frac { A }{ 3 }\)2 [B] [4L]3/2
Rate = \([\frac { 8 }{ 9 }]\) (k[A]2 [B] [L]3/2) ……………….(5)
Comparing (1) and (5); rate is reduced to 8/9 times.
![]()
Question 11.
The rate of formation of a dimer in a second order reaction is 7.5 x 10-3 mol L-1s-1 at 0.05 mol L-1 monomer concentration. Calculate the rate constant.
Solution:
Let us consider the dimensation of a monomer M
2M → (M)2
Rate = k [M]n
Given that n =2 and [M] = 0.05 mol L-1
Rate = 7.5 x 10-3 mol L-1s-1
Rate 7.5 x 103 mol L-1 s-1
k = \(\frac { Rate }{ { \left[ M \right] }^{ n } }\)
k= =\(\frac { 7.5\times { 10 }^{ -3 } }{ { \left( 0.05 \right) }^{ 2 } }\) = 3 mol-1 Ls-1
Question 12.
For a reaction x +y + z → products, the rate law is given by rate = k [x]3/2 [y]1/2 what is the overall order of the reaction and what is the order of the reaction with respect to z.
Solution:
Rate = k [x]3/2 [y]1/2
overall order = \(\left( \frac { 3 }{ 2 } +\frac { 1 }{ 2 } \right)\) = 2
i.e., second order reaction.
Since the rate expression does not contain the concentration of Z , the reaction is zero order with respect to Z.
![]()
Question 13.
Explain briefly the collision theory of bimolecular reactions.
Answer:
- Collision theory was proposed independently by Max Trautz in 1916 and William Lewis in 1918.
- According to this theory, chemical reactions occur as a result of collisions between the reacting molecules.
- Consider the reaction A2(g) + B2(g) 4 2AB(8)
- The rate of this reaction would be proportional to the number of collisions per second between A2 and B2.
Rate α number of molecules colliding per second (collision rate) - The number of collisions is directly proportional to the concentration of both A2 and B2.
Collision rate α [A2] [B2]
Collision rate = Z [A2] [B2] where Z is a constant. - Collision rate in gases can be calculated from kinetic theory of gases.
- For a gas at room temperature (298K) and 1 atm. pressure each molecule undergoes approximately 109 collisions per second. i.e. 1 collision in 10-9 second.
- Thus if every collision resulted in reaction, the reaction would he complete in 10-9 second.
- But in actual practice this does not happen.
- It implies that all collisions are not effective to lead to the reaction.
- In order to react the colliding molecules must possess minimum energy called activation energy.
- The molecules that collide with less energy than activation energy will remain intact and no reaction occurs.
- Fraction of effective collisions f = Ea/RT
- For a reaction having activation energy 100 KJ mol– at 300 K collision factor f is \(\mathrm{f}=\mathrm{e}^{-40} \cong 4 \times 10^{-18}=\frac{4}{10^{18}}\)
- Thus out of 10 collision only 4 collisions are sufficiently energetic to convert reactants to products.
- This fraction of collision is further reduced due to orientation factor.
- That is even if the reactant collide with sufficient energy, they will not react unless the orientation of the reactant molecules is suitable for the formation of the transition state.
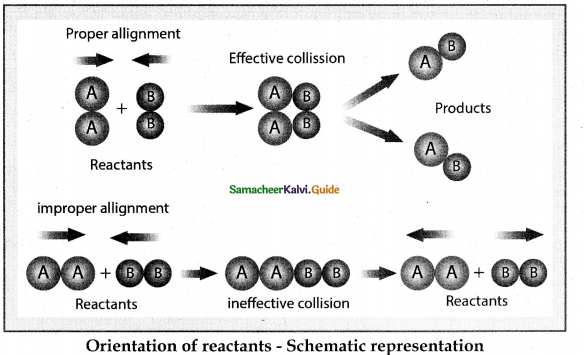
Fraction of effective collisions (O having proper orientation is given by the steric factor P.
Rate = p x f x Collision rate
Rate = pe Ea/RT [A2] [B2] …………… (1)
Rate law is Rate = k [A2] [B2] ………………..(2)
Comparing (1) & (2) k = pze Ea/RT
![]()
Question 14.
Write Arrhenius equation and explains the terms involved.
Answer:
Arrhenius equation:
k = A\({ e }^{ \frac { { -E }_{ a } }{ RT } }\)
A = Arrhenius factor (frequency factor)
R = Gas constant
k = Rate constant
Ea = Activation energy
T = Absolute temperature (in K)
Question.15.
The decomposition of Cl2O7 at 500K in the gas phase to Cl2 and O2 is a first order reaction. After 1 minute at 500K, the pressure of Cl2O7 falls from 0.08 to 0.04 atm. Calculate the rate constant in s-1.
Answer:
Solution:
k = \(\frac { 2.303 }{ t }\) log \(\frac{\left[\mathrm{A}_{0}\right]}{[\mathrm{A}]}\)
k = \(\frac { 2.303 }{ 1 min }\) log \(\frac { [0.08] }{ [0.04] }\)
k = 2.303 log 2
k = 2.303 x 0.3010
k = 0.693 2 min-1
k = \((\frac { 0.6932 }{ 60 })\) s-1
k = 1.153 x 10-2 s-1
Question 16.
Give examples for a zero-order reaction.
Answer:
Examples for a zero-order reaction:
1. Photochemical reaction between H2 and Cl
H2(g) + Cl2(g) \(\underrightarrow { h\nu }\) 2HCI(g)
2. Decomposition of N2O on hot platinum surface
N2 O(g) \(\rightleftharpoons\) N2(g) + \(\frac { 1 }{ 2 }\) O2(g)
3. iodination of acetone in acid medium is zero-order with respect to iodine.
CH3COCH3 + I2 \(\underrightarrow { { H }^{ + } } \) ICH2COCH3 + HI
Rate k [CH3COCI3] [H+]
![]()
Question 17.
Explain pseudo-first-order reaction with an example.
Answer:
A second-order reaction can be altered to a first-order reaction by taking one of the reactants in large excess, such reaction is called pseudo-first-order reaction. Let us consider the acid hydrolysis of an ester,
CH3COOCH3(aq) +H2 O(1) \(\underrightarrow { { H }^{ + } } \) CH3COOH(aq) + CH3OH(aq)
Rate = k [CH3COOCH3] [H2O]
If the reaction is carried out with a large excess of water, there is no significant change in the concentration of water during hydrolysis. i.e., the concentration of water remains almost constant. Now we can define k [H2O] = k
∴ The above rate equation becomes
Rate k [CHCOOCH] Thus it follows first-order kinetics.
Question 18.
Identify the order for the following reactions
i) Rusting of Iron
ii) Radioactive disintegration of 92U23
iii) 2A+ B → products; rate = k [A]1/2 [B]2
Answer:
i) Order of Rusting of iron.
Since Rusting of iron is a very slow reaction, it is difficult to determine its rate. Hence it is difficult to write the rate law and difficult to predict the order of rusting of iron.
ii) radioactive disintegrations 92U238 first order reactions
iii) 2A + 3B → products:
rate = k[A]1/2 [B]2
Order = \(\frac { 1 }{ 2 }\) + 2 = \(\frac { 5 }{ 2 }\) = 2.5
![]()
Question 19.
A gas phase reaction has energy of activation 200 kJ mol-1. If the frequency factor of the reaction is 1.6 x 1013 s-1. Calculate the rate constant at 600 K. (e-40.09 = 3.8 x I0-18 )
Solution:
Ea = 200KJmol-1 = 200 x 103Jmol-1
A = 1.6 x 1013s-1
T= 600K
R = 8.314Jkmol-1
e-40.09= 3.8 x 10-18
k=Ae -Ea/RT
\(=1.6 \times 10^{13} \times \mathrm{e}^{\frac{-200 \times 10^{3}}{8.314 \times 600}}\)
= 1.6 x 1013 x e-40.09
=1.6 x 1013 X 3.8 x 10-18
k = 6.08 x 10-5s-1
Question 20.
For the reaction 2x +y → L find the rate law from the following data.
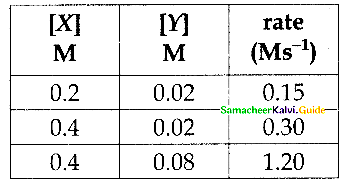
Solution:
Rate = k [x]n [y]m
0.15 = k [0.2]n [0.02]m ……………..(1)
0.30 = k [0.4]n [0.02]m ……………… (2)
1.20 = k [0.4]n [0.08]m ……………… (3)

![]()
Question 21.
How do concentrations of the reactant influence the rate of reaction?
Answer:
The rate of a reaction increases with the increase in the concentration of the reactants. The effect of concentration is explained on the basis of the collision theory of reaction rates.
According to this theory, the rate of a reaction depends upon the number of collisions between the reacting molecules. The higher the concentration, the greater is the possibility for collision and hence the rate.
Question 22.
How does the nature of the reactant influence rate of reaction?
Answer:
Rate ∝ Concentration of reactants.
Rate ∝ number of collisions between the reacting molecules.
∴ Concentration reactants a number of collisions.
As the concentration of reactants increases, the number of collisions between the reacting molecules increases, and hence the rate of reaction increases.
A chemical reaction involves breaking of certain existing bonds of the reactant and forming new bonds which lead to the product.
Net energy involved in this process is dependent on the nature of the reactant.
Hence the rates of reaction are different for different reactants.
(eg.) Redox reaction between ferrous ammonium sulphate and KMnO4 is fast and the pink colour of KMnO4 disappears immediately.
But the redox reaction between Oxalic acid and KMnO4 is relatively slow compared to the above reaction. In fact, heat is required for this reaction and the reaction is carried out at 60°C. On heating, the pink colour of KMnO4 disappears.
Hence the rate of the reaction depends on the nature of the reactant.
Question 23.
The rate constant for a first order reaction is 1.54 x 10 s-1. Calculate its half life time.
Solution:
We know that, t1/2 = 0.693 k
t1/2 = 0.693/1.54 x 10-3 = 450 s
![]()
Question 24.
The half life of the homogeneous gaseous reaction SO2Cl2 → SO2 + Cl2 which obeys first order kinetics Is 8.0 minutes. How long will it take for the concentration of SO2Cl2 to be reduced to 1% of the initial value?
Answer:
We know that, k = 0.693/ t1/2
k = 0.693/8.0 minutes = 0.087 minutes -1
For a first order reaction,
k = \(\frac { 2.303 }{ k }\) log \(\left( \frac { \left[ { A }_{ 0 } \right] }{ \left[ A \right] } \right)\)
t = \(\frac { 2.303 }{ 0.087{ min }^{ -1 } }\) log\(\frac { 100 }{ 1 }\)
t = 52.93 mm
Question 25.
The time for half change in the first-order decomposition of a substance A is 60 seconds. Calculate the rate constant. How much of A will be left after 180 seconds?
Answer:
1. Order of a reaction = 1
t1/2 = 60
seconds, k = ?
k = \(\frac { 2.303 }{ 60 }\)
We know that, k = \(\frac { 2.303 }{ { t }_{ 1/2 } }\)
k = \(\frac { 2.303 }{ 60 }\) = 0.01155 s-1
2. [A0] = 100%
t = 180 s
k = 0.01155 seconds-1
[A] = ?
For the first order reaction k = \(\frac { 2.303 }{ 60 }\) log \(\left( \frac { \left[ { A }_{ 0 } \right] }{ \left[ A \right] } \right)\)
0.9207 = log 100 – log [A]
log [A] = log 100 – 0.9207
log [A] = 2 – 0.9207
log[A] = 1.0973
[A] = antilog of (1.0973)
[A] = 12.5%
After 180 seconds 12.5% of A will be left over.
Question 26.
A zero-order reaction is 20% complete in 20 minutes. Calculate the value of the rate constant. In what time will the reaction be 80% complete?
Answer:
1. A = 100%, x = 20%, Therefore, a – x =100 – 20 = 80
For the zero order reaction k= \((\frac { x }{ t })\) ⇒
k = \((\frac { 20 }{ 20 })\) = 1
Rate constant for a reaction = 1
2. To calculate the time for 80% of completion
k = 1, a = l00, x = 80%, t = ?
Therefore, t = \((\frac { x }{ k })\) = \((\frac { 80 }{ 1 })\) = 80 min
![]()
Question 27.
The activation energy of a reaction is 225 k cal mol-1 and the value of rate constant at 40°C is 1.8 x 10-5 s-1. Calculate the frequency factor, A. Here, we arc given that
Answer:
Ea = 22.5 kcal mol-1 = 22500 cal mol-1
T = 40°C = 40 + 273 = 313 K
k = 1.8 x 10-5 sec-1
Substituting the values in the equation
log A = log k + \(\left( \frac { { E }_{ a } }{ 2.303RT } \right)\)
log A = log (l .8 x 10-5) + \(\left( \frac { 22500 }{ 2.303\times 1.987\times 313 } \right)\)
log A = log (l.8) – 5 + (15.7089)
log A = (10.9642)
A = antilog ( 10.9642)
A = 9.208 x 1010 collisions s-1
Question 28.
Benzene diazonium chloride in aqueous solution decomposes according to the equation C6H5N2CI → C6H5CI + N2. Starting with an initial concentration of 10 g L-1 volume of N2. gas obtained at 50°C at different intervals of time was found to be as under:
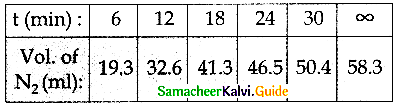
Show that the above reaction follows the first order kinetics. What is the value of the rate constant ?
Solution:
For a first order reaction
k = \(\frac { 2.303 }{ t }\) log \(\frac { a }{ (a-x) }\)
k = \(\frac { 2.303 }{ t }\) log \(\frac { { V }_{ \infty } }{ { V }_{ \infty }-{ V }_{ t } }\)
In this case, V∞ = 58.3 ml
The value of k at different time can be calculated as follows:

Since the value of k comes out to be nearly constant, the given reaction is of the first order. The mean value of k = 0.0674 min-1
![]()
Question 29.
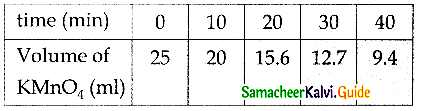
From the following data, show that the decomposition of hydrogen peroxide is a reaction of the first order:

Where t is the volume of standard KMnO4 solution required for titrating the same volume of the reaction mixture.
Solution:
Volume of KMnO4 solution used Amount of H2O2 present. Hence if the given reaction is of the first order, it must obey the equation
k = \(\frac { 2.303 }{ t }\) log \(\frac { a }{ (a-x) }\)
k = \(\frac { 2.303 }{ t }\) log \(\frac { { V }_{ 0 } }{ { V }_{ t } }\)
In this case,V0 = 46.1 ml
The value of k at each instant can be calculated as follows:

Thus, the value of k comes out to be nearly constant. Hence it is a reaction of the first order. The mean value of k = 0.04355min-1
Question 30.
A first order reaction is 40% complete in 50 minutes. Calculate the value of the rate constant. in what time will the reaction be 80% complete?
Answer:
1. For the first order reaction k = \(\frac { 2.303 }{ t }\) log \(\frac { a }{ (a-x) }\)
Assume, a = 100 %, x = 40%, t = 50 minutes
Therefore, a – x = 100 – 40 = 60
k = (2.303/50) log (100/60)
k = 0.010216 min-1
Hence the value of the rate constant is 0.010216 min-1
2. t = ?, when x = 8O%
Therefore, a – x = 100 – 80 = 20
From above, k = 0.0102 16 min-1
t = (2.303 / 0.010216) log (100 / 20)
t = 157.58 min
The time at which the reaction will be 80% complete is 157.58 min.
III. Evaluate Yourself
Question 1.
Write the rate expression for the following reactions, assuming them as elementary reactions.
i) 3A + 5B2 → 4CD
ii) X2 + Y2 → 2XY
Answer:
1. 3A + 5B2 → 4CD
Rate = – \(\frac { 1 }{ 3 }\) \(\frac { \triangle [A] }{ dt }\)
= – \(\frac { 1 }{ 5 }\) \(\frac { \triangle [{ B }_{ 2 }] }{ dt }\)
= + \(\frac { 1 }{ 4 }\) \(\frac { \triangle [CD] }{ dt }\)
2. X2 + Y2 → 2XY
Rate = – \(\frac { \triangle [{ X }_{ 2 }] }{ dt }\)
= + \(\frac { 1 }{ 2 }\) \( [latex]\frac { \triangle [{ XY }_{ 2 }] }{ dt }\)
![]()
Question 2.
Consider the decomposition of N2O5(g) to form NO2(g) and O2(g). At a particular instant N2O5 disappears at a rate of 2.5 x 10-2 mol dm-3 s-1. At what rates are NO2 and O2 formed? What is the rate of the reaction?
Solution:
2N2O5(g) → 4NO2(g) + O2(g)
from the stoichiometry of the reaction.
– \(\frac { 1 }{ 2 }\) \(\frac { d[{ N }_{ 2 }{ O }_{ 5 }] }{ dt }\)
= \(\frac { 1 }{ 4 }\) \(\frac { d[{ N }{ O }_{ 2 }] }{ dt }\)
= –\(\frac { d[{ N }{ O }_{ 2 }] }{ dt }\)
= 2 –\(\frac { d[{ N }_{ 2 }{ O }_{ 5 }] }{ dt }\)
Rate of disappearance of N2O5 is 2.5 x 10-2 mol dm-3 s-1
∴ The rate of formation of NO2 at this temperature is 2 x 2.5 x 10-2 = 5 x 10-2 mol dm-3 s-1.
– \(\frac { 1 }{ 2 }\) \(\frac { d[{ N }_{ 2 }{ O }_{ 5 }] }{ dt }\)
= – \(\frac { d[{ O }_{ 2 }] }{ dt }\)
∴ \(\frac { d[{ O }_{ 2 }] }{ dt }\) = \(\frac { 1 }{ 2 }\) x 2.5 x 10-2 mol dm-3 s-1
= 1.25 x 10-2 mol dm-3 s-1
Question 3.
For a reaction, X + Y → Product quadrupling [x], increases the rate by a factor of 8. Quailrupling both [x] and [y] increases the rate by a factor of 16. Find the order of the reaction with respect to x and y. what is the overall order of the reaction?
Solution:
X + Y → Product
Rate of Law is Rate = k[x]m[y]n
Let us take rate = 1 ∴ k[x]m [y]n …..(1)
Quailrupling [x] ie [4x], Rate = 8
∴ k[4x]m[y]n = 8
∴ km4m [x]m[y]n = 8 ……………..(2)
Quadrupling [x] and [y] ie [4x] and [4y], Rate = 16
k[4x]m[y]n = 16
k4mxm4n[y]n = 16 …………..(3)
Dividing (2) by (1)
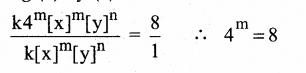
(22)m = 23
(22m ) = 23
∴ 2m = 3
m = 3/2 = 1.5
∴ Order of the reaction w.r.t. x is 1.5
Dividing (3) by (2)
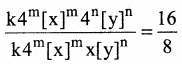
4n = 2
(22)n = 21
22n = 21
∴ 2n=1
n = 1/2
n =0.5
∴ Order of the reaction w.r.t. y is 0.5
∴ Overall order of the reaction = m + n
=1.5 + 0.5 = 2
![]()
Question 4.
Find the individual and overall order of the following reaction using the given data.
2NO(g) + Cl2 (g) → 2NOCl(g)
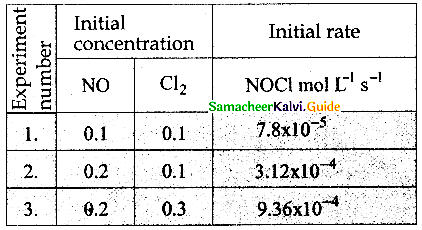
Solution:
Rate = k [NO]m [CI2]
For experiment 1, the rate law is,
Rate1 = k [NO]m [CI2]n
7.8 x 10-5 k[0.1]m [0.1]n ………………(1)
For experiment 2, the rate law is.
Rate2 = k [NO]m [CI2]n
3.12 x 10-4 = k[O.2]m [0.1]n ……………….(2)
For experiment 3, the rate law is,
Rate3 = k [NO]m [CI2]n
9.36 x 10-4 = k [O.2]m [0.3]m ……………(3)
Dividing Eq (2) by Eq (l) we get,
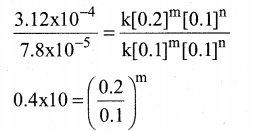
4 = \(\frac { 0.2 }{ 0.1 }\)m
⇒ 22 = 2m
∴ m = 2
Therefore the reaction is secondary order with respect to NO.
Dividing Eq (3) by Eq (2) we get,
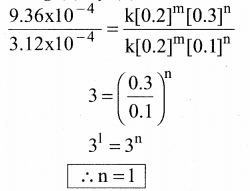
Therefore the reaction is first order with respect to Cl2
The rate law is, Rate = k [NO]2 [Cl2]1
The overall order of the reaction (2 +1) = 3.
![]()
Question 5.
In a first order reaction A → products, 60% of the given sample of A decomposes in 40 min. what is the half life of the reaction?
Solution:
k = \(\frac { 2.303 }{ t }\) log \(\frac{\left[\mathrm{A}_{0}\right]}{[\mathrm{A}]}\)
k = \(\frac { 2.303 }{ 40min }\) log \(\frac { 100 }{ (100-60) }\)
k = 0.0575 (0.3979) ⇒ k = 0.02287 min-1
t1/2 = \(\frac { 0.6932 }{ k }\) log \(\frac { 0.6932 }{ 0.02287 }\)
t1/2 = 30.31 min.
Question 6.
The rate constant for a first order reaction is 2.3 x 10-4 s-1. If the initial concentration of the reactant is 0.01 M. what concentration will remain after 1 hour?
Solution:

Question 7.
Hydrolysis of an ester in an aqueous solution was studied by titrating the liberated carboxylic acid against sodium hydroxide solution. The concentrations of the ester at different time intervals are given below.

Show that, the reaction follows first order kinetics.
Solution:

Since all the k values are constant
This value shows that reaction follws first order kinetics.
![]()
Question 8.
For a first order reaction the rate constant at 500K is 8 x 10-4 s-1. Calculate the frequency factor, if the energy of activation for the reaction is 190 kJ mol-1.
k = 8 x 10-4s
T = 500K
Ea = 190 kJ mol-1 A = ?
Solution:

12th Chemistry Guide Chemical Kinetics Elements Additional Questions and Answers
I. Match the following
Question 1.
| Rate constant | Unit |
| i. Zero order reaction | a. mol-2L-2s-1 |
| ii. First order reaction | b. molL-1s-1 |
| iii. Second order reaction | c. s-1 |
| iv. Third order reaction | d. mol-1Ls-1 |
Answer:
i. b. molL-1s-1
ii. c. s-1
iii. d. mol-1Ls-1
iv. a. mol-2L-2s-1
Question 2.
| I | II |
| I Catalyst | a. can be negative |
| ii Order | b. independent of initial concentration |
| iii Molecularity | c. lowers the activation energy |
| iv Half life of first order reaction | d. can not be fraction |
Answer:
i) c. lowers the activation energy
ii) a. can be negative
iii) d. can not be fraction
iv) b. independent of initial concentration
II. Assertion and Reason
i) Both A and R are correct and R explains A.
ii) A is correct but R is wrong.
iii) A is wrong but R is correct.
iv) Both A and R are correct but R does not explain A.
Question 1.
Assertion (A): Rate of a reaction can be
\(\text { written as }-\frac{\mathrm{d}[\text { Reactant }]}{\mathrm{dt}}\)
Reason (R): As time increases concentration of reactant decreases.
Answer:
(i) Both A and R are correct and R explains A.
![]()
Question 2.
Assertion (A) : For a gas phase reaction the unit of reaction rate is atm s-1
Reason (R) : For a gas phase reaction, the concentration of the gaseous species is expressed in terms of mole per litre.
Answer:
(ii) A is correct but R is wrong.
Correct R: For a gas phase reaction, the concentration of the gaseous species is expressed in atm.
Question 3.
Assertion (A) : For a general reaction xA + yB → products, rate law is Rate = k[A]m [B]n
Reason (R) : The values of m and n can be deduced from the stoichiometry of the reaction.
Answer:
(ii) A is correct but R is wrong.
Correct R: The values of m and n can be deduced from experiments.
Question 4.
Assertion (A) : Acid hydrolysis of an ester is a pseudo first order reaction.
Reason (R) : The rate of the reaction does not depend on the concentration of one of the reactants water.
Answer:
i) Both A and R are correct and R explains A.
III. Choose the correct statements.
Question 1.
(i) During the reaction the concentration of the reactant increases.
(ii) Unit of rate = Unit of concentration (iii) For a gas phase reaction the unit of rate is atm s-1
(a) (i) & (ii) (b) (i) & (iii) c) (ii) &(iii) d) (iii) only
Answer:
(d) (iii) only
Correct statement: (i) During the reaction the concentration of the reactant decrease.
Unit of concentration Unit of time
![]()
Question 2.
(i) The rate of a reaction decreases with time as the reaction proceeds.
(ii) The average rate can be used to predict the rate of the reaction at any instant.
(iii) The rate of the reaction at a particular instant during the reaction is called the instantaneous rate.
(a) (i) & (ii)
(b) (i) & (iii)
c)) (ii) & (iii)
(d) (i), (ii) & (iii)
Answer:
(b) (i) & (iii)
Correct statement:
(ii) The average rate cannot be used to predict the rate of the reaction at any instant.
Question 3.
(i) Rate constant is a proportionality constant
(ii) Rate of a reaction depends on the initial concentration of the reactants.
(iii) Rate constant does not depend on the initial concentration of the reactants.
(a) (i) & (ii)
(b) (i) & (iii)
(c) (ii) & (iii)
(d) (i), (ii) & (iii)
Answer:
(d) (i), (ii) & (iii)
![]()
Question 4.
(i) Order is assigned for each elementary step of mechanism.
(ii) Order can be fractional.
(iii) Molecularity can not be fractional.
(a) (i) & (ii)
(b) (i) & (iii)
(c) (ii) & (iii)
(d) (i), (ii) & (iii)
Answer:
(c) (ii) & (iii)
Correct statement: (i) Molecularity is assigned for each elementary step of mechanism.
IV. Choose the incorrect statements.
Question 1.
(i) All radio active reactions are first order
reaction.
(ii) Half life of first order reaction depends on the initial concentration of the reactants.
(iii) For a first order reaction a plot of ln[A] against t gives a straight line with positive slope.
a) (i) & (ii)
b) (i) & (iii)
c) (ii) & (iii)
d) (i), (ii) & (iii)
Answer:
(c) (ii) & (iii)
Correct statement:
(ii) Half life of first order reaction does not depend on the initial concentration of the reactants.
(iii) For a first order reaction a plot of ln[A] against t gives a straight line with negative slope.
![]()
Question 2.
(i) Acid hydrolysis of an ester is a second order reaction.
(ii) Iso merisation of cyclo propane to propene is a zero order reaction.
(iii) In acid hydrolysis of ester the concentration of ester remains constant.
a) (i) & (ii)
b) (i) & (iii)
c) (ii) & (iii)
d) (i), (ii) & (iii)
Answer:
d) (i), (ii) & (iii)
Correct statement:
(i) Acid hydrolysis of an ester is a Pseudo first order reaction
(ii) Isomerisation of cyclo propane to propene is a first order reaction.
(iii) In acid hydrolysis of ester the concentration of water remains constant.
Question 3.
(i) A zero order reaction is independent of the
concentration of the reactants.
(ii) Iodination of acetone in acid medium is zero order with respect to acetone.
(iii) Zero order reactions are quite common.
(a) (i) & (ii)
(b) (i) & (iii)
(c) (ii) & (iii)
(d) (i), (ii) & (iii)
Answer:
(c) (ii) & (iii)
Correct statement:
(ii) Iodination of acetone in acid medium in zero order with respective to iodine.
(iii) Zero order reaction are rare.
![]()
Question 4.
(i) Rate of a reaction is inversely propor-tional
to the number of collisions per second.
(ii) Collision rate in gases can be calculated from kinetic theory of gases.
(iii) Number of collisions is inversely proportional to the concentration of the reactants.
(a) (i) & (ii)
(b) (i) & (iii)
(c) (ii) & (iii)
(d) (i), (ii) & (iii)
Answer:
(b) (i) & (iii)
Correct statement:
(i) Rate of a reaction is directly proportional to the number of collision per second.
(iii) Number of collisions is directly proport¬ional to the concentration of the reactants.
V. Choose the best Answer
Question 1.
As the reaction proceeds the concentration of the reactant
a) increases
b) decreases
c) remains the same
d) tends to be maximum
Answer:
b) decreases
![]()
Question 2.
For a general reaction xA + yB → Ic + mD regarding its rate which is incorrect

Answer:
b
Question 3.
The rate constant of a reaction is 2.3 x 10-2 lit mol-1 S-1
The order of the reaction is
a) Zero order
b) First order
c) Second order
d) Third order
Answer:
c) Second order
Question 4.
The sum of exponential terms in the rate law is called as
a) Molecularity
b) Rate constant
c) Order
d) Rate
Answer:
c) order
Question 5.
The value of order of a reaction can be
a) Zero
b) Fractional
c) Integer
d) all the above
Answer:
d) all the above
![]()
Question 6.
The value of molecularity of a reaction can be
a) Zero
b) Fractional
c) whole number
d) all the above
Answer:
c) whole number
Question 7.
During the decomposition of H2O2 (H2O2 → 2H2O + O2). 48g of O2 is formed per Minute at particular instant. The rate of formation of water at this instant in mol min-1 is
a) 1
b) 2
c) 3
d) 4
Answer:
c) 3
Question 8.
If the rate law of a reaction is Rate = k[A]3/2[B]1 the order of the reaction with respect to A is
a) 1.5
b) 1
c) 2.5
d) 3
Answer:
a) 1.5
![]()
Question 9.
If the rate law of a reaction is Rate = k[A]3/2[B]1 the order of the reaction with respect to B is
a) 1.5
b) 1
c) 2.5
d) 3
Ans:
b) 1
Question 10.
If the rate law of a reaction is Rate = k[A]3/2[B]1 the overall order of the reaction is
a) 1.5
b) 1
c) 2.5
d) 3
Answer:
c) 2.5
Question 11.
If the rate law for a reaction is Rate = k[A]2 [B]°, the unit of rate constant k for the
overall reaction is
(a) molL-1V
(b) mol-1LS-1
(c) mol-2L-2S-1
(d) S-1
Answer:
(b) mol-1LS-1
Question 12.
Which among the following is the unit of k of a zero order reaction ?
(a) mol-1L-1S-1
(b) mol-1LS-1
(c) mol-2L-2S-1
(d) S-1
Answer:
(a) mol-1L-1S-1
![]()
Question 13.
The rate law of a reaction is Rate = k[A] [B], if the concentration of B is taken in large excess, the order of the reaction is
a) zero order
b) first order
c) pseudo first order
d) second order
Answer:
c) pseudo first order
Question 14.
The rate determining step of a reaction is ……………. step.
a) fast
b) slow
c) equilibrium
d) intermediate
Answer:
b) slow
Question 15.
From the following which is a second order rate constant?
a) k = 5.47 x 10-4s-1
b) k = 3.9 x 10-3mol lit s-1
c) k = 3.94 x 10-4 lit mol -1 s-1
d) k = 3.98 x 10-5 lit mol-2s-1
Answer:
c) k = 3.94 x 10-4 lit mol -1 s-1
Question 16.
Total number of reactant species that are involved in an elementary step is called as
a) order
b) molecularity
c) rate
d) rate constant
Answer:
b) molecularity
![]()
Question 17.
For an elementary reaction 2A + B → C + D the molecularity is
a) zero
b) one
c) two
d) three
Answer:
d) three
Question 18.
Which is correct for first order reactions,
a) t 1/2 ∝ (concn)-1
b) t 1/2 ∝ (concn)
c) t 1/2 ∝ (concn)0
d) t 14/2 ∝ (concn)1/2
Answer:
c)
Question 19.
Time required for the reactant concentration to reach one half of its initial value is called
a) Half life period
b) First order
c) zero order
d) Second order
Answer:
a) Half life period
Question 20.
Molecularity can be determined by
a) Stoichiometry
b) experiment
c) mechanism
d) none of the above
Answer:
c) mechanism
![]()
Question 21.
Rate constant of a reaction is equal to the rate of the reaction when the concentration of the reactants is
a) zero
b) unity
c) infinity
d) fractional
Answer:
b) unity
Question 22.
For a first order reaction a plot of /n[A] against Y yields a straight line with a slope which is
a) positive
b) negative
c) zero
d) at infinity
Answer:
b) negative
Question 23
Which of the following is not a first order reaction ?
i) Decomposition of dinitrogen pentoxide.
ii) Decomposition of N20 on hot platinum surface.
iii) Decomposition of thionyl chloride.
a) (i) only
b) (i) & (ii)
c) (ii) only d) (ii) & (iii)
Answer :
c) (ii) only
Question 24.
Which of the following are zero order reaction ?
i) Isomerisation of cyclo propane to propene.
ii) Iodination of acetone in acid medium with respect to Iodine.
iii) Photo chemical reaction between H2 and Cl2
a) (i) only
b) (i) & (ii)
c) (ii) only
d) (ii) & (iii)
Answer:
d) (ii) & (iii)
![]()
Question 25.
Acid hydrolysis of an ester is an example for ……………. order reaction.
a) zero
b) first
c) pseudo first
d) second
Answer:
c) pseudo first
Question 26.
Base hydrolysis of an ester is an example for ………….. order reaction
a) zero
b) first
c) pseudo first
d) second
Answer:
d) Second
Question 27.
The half life period of a first order reaction is
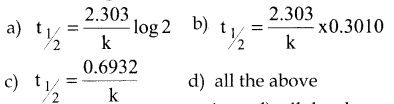
Answer:
d) All the above
![]()
Question 28.
If the initial concentration of the reactant is doubled, the half life period of a first order reaction is
a) doubled
b) tripled
c) quadrupled
d) remains the same
Answer:
d) remains the same
Question 29.
The half life period of a zero order reaction is
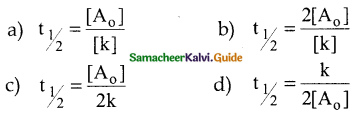
Answer:
c)
Question 30.
If tiie initial concentration of the reactant is doubled, the half life period of a zero order reaction is
a) doubled
b) tripled
c) quadrupled
d) remains the same
Answer:
a) doubled
Question 31.
The half life period of a second order reaction is
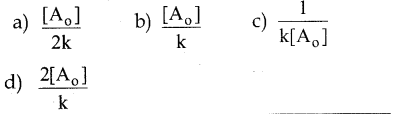
Answer:
c)
![]()
Question 32.
The half life period of a first order reaction is 5 minutes, the time required for 99.9% completion is nearly equal to
a) 99.9 minu tes
b) 49.95 minutes
c) 50 minutes
d) 10 minutes
Answer:
c) 50 minutes
Question 33.
The time required for 99.9% completion of a first-order reaction is equal to
a) 2t1/2
b) 5t1/2
c) 10t1/2
d) 100t1/2
Answer:
c) 10t1/2
Question 34.
Collision theory was proposed independently by
a) MaxTrautz
b) William Lewis
c) both (a) & (b)
d) none of the above
Answer:
c) both (a) & (b)
Question 35.
Collision theory is based on
a) Arrhenius theory
b) Kinetic theory of gases
c) Ostwald theory
d) Gas laws
Answer:
b) Kinetic theory of gases
![]()
Question 36.
In order to react the colliding molecules must possess a minimum energy called
a) Kinetic energy
b) activation energy
c) potential energy
d) bond energy
Answer:
b) activation energy
Question 37.
The rate of a reaction ………… with increasing temperature
a) increases
b) decreases
c) becomes zero
d) remains the same
Answer:
a) increases
Question 38.
For many reactions near room temperature, reaction rate tends to …………….. when the temperature is increased by 10 C.
a) decreases
b) double
c) triple
d) quaruple
Answer:
b) double
Question 39.
The slope for the straight line obtained from the plot of Ink Vs 1/T is T
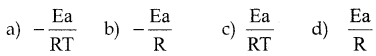
Answer:
b)
![]()
Question 40.
The y intercept of the straight line obtained from the plot of Ink vs 1/T is
a) In k
b) \(-\frac{\mathrm{Ea}}{\mathrm{R}}\)
c) In A
d) \(-\frac{\mathrm{Ea}}{\mathrm{RT}}\)
Answer:
c) In A
Question 41.
Rate of a reaction …………….. with increase in concentration of the reactants
a) decreases
b) increases
c) remains the same
d) is slower
Answer:
b) increases
Question 42.
As concentration of the reactant increases the number of collisions between the molecules
a) decreases
b) increases
c) rem a ins the same
d) is slower
Answer:
b) increases
Question 43.
Increase in surface area of the reactant…………….the rate of the reaction
a) increases
b) decreases
c) no change
d) is slower
Answer:
a) increases
Question 44.
Increase in surface area of the reactant ………….. the number of collisions between the molecules.
a) increases
b) decreases
c) no change
d) is slower Ans :
a) increases
![]()
Question 45.
Generally addition of a catalyst ……………. the rate of a reaction
a)increases
b)decreases
c) inhibits
d) has no change
Answer:
a) increases
Question 46.
A catalyst ……………. the activation energy of a reaction.
a) increases
b) decreases
c) has no change
d) none of the above
Answer:
b) decreases
Question 47.
What is the rate law of the reaction 2A + 2B → C + 2D. If the concentration of A is doubled at constant [B] the rate of the reaction increases by factor 4. If the concentration of B is doubled at constant [A], the rate is doubled.
a) Rate = k [A] [B]2
b) Rate = k [A] [B]
c) Rate = k [A] 1/2 [B]2
d) Rate = k [A]2[B]
Answer:
d) Rate = k [A]2[B]
Question 48.
The concentration of a reactant decreases from 0.5 M to 0.3 M in 10 minutes. The rate of the reaction is
a) 0.01mol L-1 S-1
b) 0.02 mol L-1 S-1
c) 0.08mol L-1 S-1
d) 0.15mol L-1 S-1
Answer:
b) 0.02 mol L-1 S-1

![]()
Question 49.
The rate of a reaction is doubled for every 10°C rise in temperature. The increase in reaction rate as a result of temperature rise from 10 C to 50°C is
a) 4
b) 8
c) 16
d) 32
Answer:
c) 16 Increase in reaction rate = 2No 10°C rise
Froml0°C to 10°C = 4 times 10°C rise
∴ 24 = 16
Question 50.
For the reaction 2N2o5 → 4N02 + O2
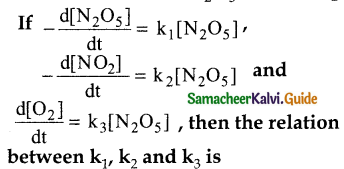
a) k1 = k2 = k3
b) k1 = 2k2 = 4k3
c) 2k1 = 4k2 = k3
d) 2k1 = k2 = 4k3
Answer:
d) 2k1 = k2 = 4k3
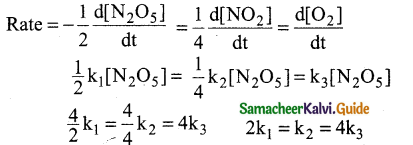
![]()
Question 51.
Rate law of a reaction A + B → C is Rate = k[A] [B] . If the concentration A and B are doubled at constant volume then the rate of the reaction will be increased by
a) two times
b) four times
c) eight times
d) sixteen times
Answer:
c) eight times
Rate = k[A][B]2
[A] & [B] are doubled
Rate = k[2A][2B]2
Rate = k2[A]22[B]2
Rate = 8 x k[A][B]2
Question 52.
If 60% of a first order reaction is complete in 60 minutes, its half life period is approximately (log4 = 0.6)
a) 50 minutes
b) 45 minutes
c) 60 minutes
d) 40 minutes
Answer:
b) 45 minutes

![]()
Question 53.
Half life period of a first order reaction is 1386 seconds. The rate constant of the reaction is
a) 5.0 x 10-3s-1
b) 0.5 x 10-2s-1
c) 0.5 x 10-3s-1
d) 5.0 x 10-2s-1
Answer:
c) 0.5 x 10-3s-1
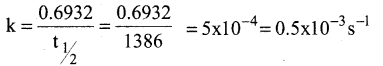
Question 54.
For the reaction 2A + B → 3C + D
Which of the following does not express the reaction rate?

Answer:
c
Question 55.
The half life period of a first order reaction is 10 minutes. If the initial concentration is 0.08 mol L-1 at what time the concentration will become 0.01 mol L -1
a) 10 minutes
b) 20 minutes
c) 30 minutes
d) 40 minutes
Answer:
c) 30 minutes
t1/2 = 10minutes

Question 56.
For a reaction the rate law is Rate = k [A]3/2[B]-1/2 , the over all order of reaction is
a) 2
b) 1
c) -1/2
d) 3/2
Answer:
b) 1
![]()
Question 57.
A first order reaction is half completed in 45 minutes . How long does it need 99.9% of the reaction to be completed?
a) 5 hours
b) 7.5 hours
c) 10 hours
d) 20 hours
Answer:
b) 7.5 hours
t99.9% = 10t1/2 = 10 x 45
= 450 minutes = 450/60 = 7.5hours
Question 58.
If the activation energy for a reaction at TK is 2.303 RT J mol-1, the ratio of rate constant to frequency factor is
a) 2 x 10-3
b) 2 x 10-2
c) 10-1
d) 10-2
Answer:
c) 10-1
logk = log A – \(\frac{\mathrm{Ea}}{2.303 \mathrm{RT}}\)
l°gk = logA – \(\frac{2.303 \mathrm{RT}}{2.303 \mathrm{RT}}\)
logk = log A – 1
logk – log A = -1
\(\log _{10}\left(\frac{\mathrm{k}}{\mathrm{A}}\right)=-1\)
\(\therefore\left(\frac{\mathrm{k}}{\mathrm{A}}\right)=10^{-1}\)
Question 59.
Chemical reactions with very high Ea values are generally
a) very fast
b) very slow
c) moderately fast
d) spontaneous
Answer:
b) very slow
![]()
Question 60.
If the activation energy of a reaction is zero, the rate constant of this reaction
a) increases with increase in temperature
b) decreases with increase in temperature
c) decreases with decrease in temperature
d) is independent of temperature
Answer:
d)is independent of temperature
Ea = 0; k = \(\mathrm{Ae}^{\frac{-\mathrm{Ea}}{\mathrm{RT}}}\)
k = \(\mathrm{Ae}^{\frac{-\mathrm{O}}{\mathrm{RT}}}\)
k = Ae°
k = A.
Hence k is independent of temperature
VI. Two mark Questions
Question 1.
Define rate of a reaction.
Answer:
The change in concentration of a species involved in a chemical reaction per unit time is called the rate of the reaction.
![]()
Question 2.
Write the rate expression of the following reaction.
N2 + 3H2 → 2NH3
Answer:
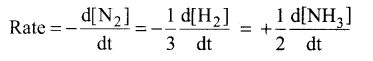
Question 3.
Why a negative sign is introduced in the rate expression ?
Answer:
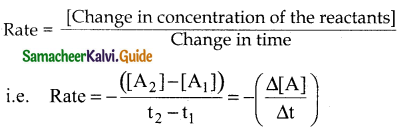
During the reaction, the concentration of the reactant decreases ie. [A2] < [A1] and hence the change in concentration [A2] – [A1] gives a negative value. By convention the reaction rate is positive one and hence a negative sign is introduced in the rate expression.
Question 4.
Define Order of a reaction.
Answer:
Order of a reaction is the sum of powers of concentration terms involved in the experimentally determined rate law.
Question 5.
Define molecularity of a reaction.
Answer:
Molecularity of a reaction is the total number of reactant species that are involved in an elementary step of a reaction.
![]()
Question 6.
Mention the factors that affect the rate of the reaction.
Answer:
- Nature and state of the reactant
- Concentration of the reactant
- Surface area of the reactant
- Temperature of the reaction
- Presence of a catalyst.
VII. Three mark Questions
Question 1.
Write the differences between rate and rate constant of a reaction.
Answer:
Rate of a reaction :
- It represents the speed | at which the reactants I are converted into products at any instant.
- It is measured as decre-ase in the concentration of the reactants or increase in the concentration of products.
- It depends on the initial concentration of reactants.
Rate constant of a reaction:
- It is a proportionality constant.
- It is equal to the rate of reaction, when the concentration of each of the reactants is unity.
- It does not depend on the initial concentration of reactants.
![]()
Question 2.
Give three examples for first order reaction.
i) Decomposition of dinitrogen pentoxide
N2O5(g) → 2NO2(g) + \(\frac{1}{2}\)O2(g)
ii) Decomposition of sulphuryl chloride
SO2Cl2(I) → SO2(g) + Cl(g)
iii) Decomposition of hydrogen peroxide in aqueous solution
H2O2( aq) → H2O2(I) + \(\frac{1}{2}\)O2
![]()
Question 3.
Calculate the half -life period of zero order reaction.
Answer:
Let us calculate the half life period for a zero order reation.

Question 4.
For the general reaction A → B, Plot of concentration of A Vs time is given in the graph below.
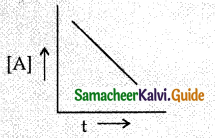
Answer the following questions on the basis of this graph.
(i) What is the order of reaction?
(ii) What is the slope of the curve?
(iii) What is the unit of rate constant?
Answer:
(i) It is a zero order reaction as the graph is satisfying the equation [A] = [A0] – kt
(ii) The slope of the curve is the negative of the rate constant that is denoted bv -k.
(iii) Unit of rate constant is Ms-1 or mol L-1 s-1
![]()
VIII.Five mark Questions
Question 1.
Derive the integrated rate law for a first order reaction.
Answer:
Let us consider a first order reaction.
A → products
The rate of first order reaction depends on the reactant concentration raised to the first power.
∴ Rate law can be written as. Rate = k [A]1
k is the first order rate constant
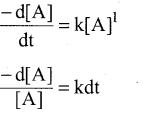
Integrating between the limits t = 0 to t t [A]
and concentration [A0] to [A]
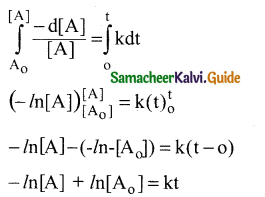
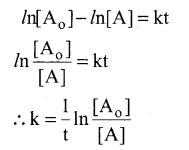
Converting natural logarithm to usual logarithm
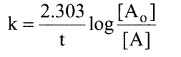
Question 2.
Derive Arrhenius equation to calculate activation energy from the rate constant k1 and k2 temperature T1 and T2 respectively.
Answer:
- The rate of reaction generally increases with increase in temperature.
- For many reactions near room temperature, reaction rate tends to double when the temperature is increased by 10°C.
- Arrhenius suggested that the rates of most reactions vary with temperature.
He proposed that the rate constant is directly proportional to e RT
\\(\mathrm{e} \frac{-\mathrm{Ea}}{\mathrm{RT}}\)
k ∝ \(\mathrm{e} \frac{-\mathrm{Ea}}{\mathrm{RT}}\)
k = A\(\mathrm{e} \frac{-\mathrm{Ea}}{\mathrm{RT}}\)
k = Rate constant
A = Frequency factor
Ea = activation energy
R = Gas constant
T = Temperature in KelvinFrequency factor A is related to the frequency of collisions between the reactant molecules.
- Frequency factor A does not vary significantly with temperature and hence it may be taken as a constant.
- Ea the activation energy is the minimum energy that a molecule must have to posses to react.
k = A\(\mathrm{e} \frac{-\mathrm{Ea}}{\mathrm{RT}}\) ……… (1)
Taking logarithm Ln k = lnA+ lne \(\mathrm{e} \frac{-\mathrm{Ea}}{\mathrm{RT}}\)
lnk= ln A – \(\frac{-\mathrm{Ea}}{\mathrm{RT}}\) (∵ lne = 1)
T = T1 , k = k1
T = T2 , k = k2

This equation can be used to calculate F.a from rate constant k1 and k2 at temperature T1 and T2
![]()
Question 3.
Explain the factors affecting reaction rate.
Answer:
i) Nature and state of the reactant.
- A chemical reaction involves breaking of certain existing bonds of the reactant and forming new bonds which lead to the product. The net energy involved in this process is dependent on the nature of the reactant and hence the rates are different for different reactants.
- (eg)- Redox reaction between ferrous ammonium sulphate and KMnO4 is fast and the pink colour of KMnO4 disappears on cold.
- Redox reaction between oxalic acid and KMn04 is slow and on heating the reaction proceeds faster and the pink colour of KMn04 disappears on heating.
- Physical state of the reactant plays an important role on the rate of the reaction. Gas phase reactions are faster as compared to the reactions involving solid or liquid reactants.
- (eg). Sodium metal with iodine vapours react faster than solid sodium and solid iodine. Solid lead nitrate and solid potassium iodide react slowly than aqueous solution of both which gives yellow precipitate immediately.
ii) Concentration of the reactants.
- Rate of the reaction increases with increase in the concentration of the reactants.
- This can be explained on the basis of collision theory of reaction rates.
- Rate of reaction depends upon the number of collisions between the reacting molecules.
- As the concentration increases, the number of collisions increases and hence rate increases.
iii) Effect of surface area of the reactant.
- When the particle size decreases surface area increases.
- Increase in surface area of reactant leads to more collisions per litre per second.
- Hence the rate of reaction is increased.
- (eg) Powdered calcium carbonate reacts much faster with dilute HC1 than with the same mass of CaCO3 marble.
iv) Effect of temperature
- The rate of reaction generally increases with increase in temperature.
- For many reactions near room temperature reaction rate tends to double when the temperature is increased by 10°C.
- (cg) Magnesium reacts readily with hot water resulting in a basic solution indicated by the pink colour of phcnolphthalein than cold water.
v) Effect of Catalyst.
- A catalyst is a substance which alters the rate of a reaction without itself undergoing any permanent chemical change.
- A catalyst may participate in the reaction, but again regenerated at the end of the reaction.
- A catalyst lowers the activation energy hence greater number of molecules can cross the energy barrier and change over to products. Thus the rate of the reaction is increased.
![]()
IX. Problems based on rate constant
Question 1.
From the following data on N2O5 decomposition in CCl4 at 298K show that the reaction is first order. Also evaluate the rate constant of the reaction.
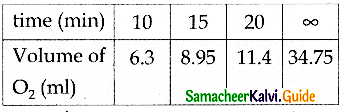
Solution:
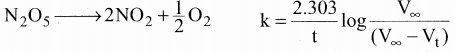

Since k values are nearly constant for different timings, this is a first order reaction.
Question 2.
From the following data, show that decomposition of H2O2 in aqueous solution follows first order reaction, what is the value of rate constant ?
Answer:

Since all the k values are nearly constant the reaction is first order.
![]()
Question 3.
In a first order reaction, it takes the reactant 40.5 minutes to be 25% decomposed. Calculate the rate constant
of the reaction.
Solution:
[A0] = 100%
[A] = 100 – 25 = 75%
t = 40.5 mins
k = ?

Question 4.
The rate of formation of a dimer in a second order reaction is 7.5 x 10-3 mol L-1S-1 at 0.05 mol L-1 monomer concentration. Calculate the rate constant.
Solution:
Let us consider the dimerisation of a monomer M
2M → (M)2
Rate = k[M]n
Given that n = 2 and [M]=0.05 mol L-1
Rate = 7.5 x 10-3mol L-1 S-1
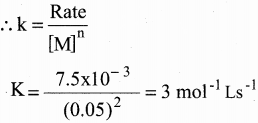
![]()
X. Problems based on half life period
At 25°C the rate constant of a first order reaction is 0.45 s-1. What is its half life? Calculate the time required for 12.5% of the reactant to remain.
Solution:
k = 0.45 s-1
t1/2 = ?
\(\mathrm{t}_{1 / 2}=\frac{0.6932}{\mathrm{k}}=\frac{0.6932}{0.45}=1.54 \mathrm{sec}\)

time needed to 12.5% of reactant to remain= 3t1/2 = 3 x 1.54 = 4.62 sec
Question 2.
The rate constant of a first order reaction is 1.54 x 10-3 s-1. What is its half life?
k = 1.54 x 10-3 sec-1, t1/2 = ?
Solution:
t1/2 = \(\frac{0.6932}{1.54 \times 10^{-3}}\) = 450 sec
Question 3.
The half life period of a first order reaction is 10 mins, what percentage of the reactant will remain after one hour?
Solution:
t1/2 = 10mins
t = 1hour = 60 mins = 6×10 min = 6 x t1/2
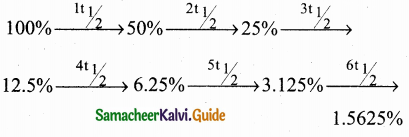
After one hour 1.5625% of the reactant will remain.
![]()
Question 4.
A first order reaction is 25% complete in 100 minutes. Calculate its rate constant and half life.
Solution:
t =100mins
[A0] = 100%
[A] = 100 – 25 = 75%
k = ?
t1/2 = ?

Question 5.
75% of a first order reaction is completed in 48 minutes. What is its half life ?
Solution:
t = 48 mins
[A0] =100%
[A] = 100-75 = 25% k = 25%
k = ?
t1/2 = ?


![]()
Question 6.
Show that for a first order reaction the time required for 99% completion of the reaction is twice the time required for 90% completion of the reaction.
Solution:

![]()
XI. Problems based on activation energy
Question 1.
For a first order reaction if the rate constant at 25°C is 3.46 x 10-5 s-1 and the rate constant at 35°C is 13.50 x 10-5 s-1, calculate the activation energy Ea and frequency factor A.
Solution:
T1 = 25°C + 273 = 298K
k1 = 3.46 x 10-5s-1
T1 = 35°C + 273 = 308K
k1 = 3.46 x 10-5s-1

= log3.46 x log 10-5 + 18.21
= log 3.46 + log 10-5 + 18.21
= Iog3.46 – 51og10 + 18.21
= 0.5391-5 + 18.21
logA = 13.7491
A = Anti log 13.7491
A = 5.611 x 1013
![]()
Question 2.
The activation energy of a certain reaction is 100 kj mol-1. What is the change in the rate constant of the reaction if the temperature is changed from 25°C to 35°C ? Let the rate constants at 25°C be k1 and at 35°C be k2 respectively.
Answer:
T1 = 25°C + 273 = 298K =>K = k1
T2 = 35°C + 273 = 308K =>K = k2
Ea = 100kjmol-1 = 100 x 103 Jmol-1
R = 8.314Jk-1mol-1

∴ k2, the rate constant at 35°C will be 3.70 times the rate constant k1 at 25°c.
Question 3.
Decomposition of ethyl bromide and propyl bromide follow first order kinetics and have the same frequency factor ‘A’. The rate constant for decomposition of ethyl bromide at 390°C is same as that for propyl bromide at 320’C. If Ea of ethyl bromide reaction is 230 kj mol-1. What is the Ea of propyl bromide reaction?
Solution:
log k = log A – \(\frac{\mathrm{E}_{\mathrm{a}}}{2.303 \mathrm{RT}}\)
For ethyl bromide decomposition at 390°C ie 663k
log k = log A – \(\frac{230 \times 10^{3}}{2.303 \times 8.314 \times 663}\) ………….(1)
For propyl bromide decomposition at 320°C ie 593k
logk = logA – \(\frac{\mathrm{E}_{\mathrm{a}}}{2.303 \times 8.314 \times 593} …………………. (2)
Since k values for both reactions are equal equation (1) & (2) are equal.

![]()
Question 4.
The rate constant k values of a reaction were determined at several temperatures. A plot of In k vs 1/T gave a straight line with the slope – 2.6 x 104 K. What is the activation energy Ea of the reaction?
Solution:
slope = [latex]-\frac{E_{a}}{R}\)
Ea = -Rslope
= -8.3 14 x (-2.6 x 104)
= 21.61 x 104 Jmol-1
Ea = 216.1 kJmol-1
Question 5.
The rate constant for a first order reaction at 45°C is twice that at 35°C. Find the activation energy of the reaction.
At T1 = 35°C + 273 = 308K
rate constant k1
T2 =45°C + 273 = 318K rateconstanf k2 =2k1
R= 8.3 14Jk-1 mol-1 Ea = ?
Solution:
